Multivisceral resections for rectal cancers: short-term oncological and clinical outcomes from a tertiary-care center in India
Introduction
Rectal cancer is the third most common cancer worldwide with an estimated age-standardized rate between 4.1–5.8 and 3.5–5.3 per 100,000 in male and in female subjects, respectively (1). Neoadjuvant chemoradiotherapy (NACTRT) followed by total or partial tumor-specific mesorectal excision remains the mainstay of treatment for locally advanced rectal cancers (LARCs) (2). Circumferential resection margin (CRM) positivity is the single most important factor predicting local recurrence and overall prognosis after rectal cancer surgery (3,4). As a result, complete surgical resection remains the primary goal of rectal cancer surgeries. MERCURY study group did show that CRM involvement on magnetic resonance imaging (MRI) is a better predictor of disease control than AJCC TNM stage; this is applicable for standard rectal cancer surgeries after neoadjuvant therapy (5). Multivisceral resections (MVRs) remove the involving adjacent organ and, hence, may lead to superior oncological outcomes in spite of involvement of CRM on MRI.
In upto 10–20% patients, primary tumor is adherent to adjacent organs because of either direct infiltration or peritumoral inflammatory reaction. However, neither preoperative nor intra-operative assessment is adequate to distinguish these adhesions as benign or malignant. Hence, en bloc resection of the tumor along with the involved structures is essential to achieve negative resection margins (R0) and to prevent spillage of tumor cells into the peritoneal cavity (6). Encouraging oncological outcomes have been achieved, with such MVRs with 5-year overall survival (OS) rates of upto 36–53% (7,8). These MVRs are often technically difficult, anatomically extensive, and associated with a high level of morbidity (9). There is dearth of Indian literature on the role of MVR for rectal cancers. The aim of this study was to determine the feasibility and safety of MVR for rectal cancer and, hence, to determine the patients who would benefit the most from such aggressive resections.
Methods
This is a retrospective review of a prospectively maintained database in the Division of Colorectal Surgery at the Tata Memorial Centre, Mumbai, Maharashtra, India. Between 1 July 2013 and 31 May 2015, all patients undergoing MVR for adenocarcinoma of the rectum were identified from this database. All patients who underwent en bloc resection of an adjacent organ or part of an adjacent organ were included. Exclusion criteria included those with unresectable metastatic disease after neoadjuvant therapy.
Following a detailed history and physical examination, all patients underwent a complete colonoscopy with biopsy and determination of serum carcino embryonic antigen (CEA) levels. Loco regional staging was performed with a baseline MRI pelvis (MRI 1) in all patients (Figure 1). Contrast-enhanced computed tomography (CECT) of the thorax and abdomen were performed to rule out distant metastasis. NACTRT was administered to all patients with a threatened CRM and/or enlarged mesorectal nodes. All treatment decisions were taken by a multidisciplinary team comprising a colorectal surgeon, a radiation oncologist, a gastroenterologist, and a radiologist. Response assessment MRI (MRI 2) was performed after 6–10 weeks of completion of NACTRT, just before definitive surgery. For all the patients who required exenteration, positron emission tomography-CT was performed in addition to the routine workup. Those patients who had threatened CRM even after NACTRT received further chemotherapy before surgery.
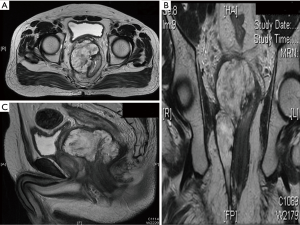
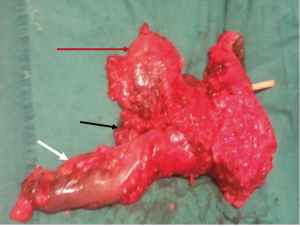
Patients were selected for MVR based on contiguous pelvic organ invasion on MRI 2 in the absence of incurable metastatic disease. However, patients with operable liver metastases were still offered MVR. Informed, written consent was obtained from all patients with extensive preoperative counseling about the procedure, its morbidity, and consequences. Rectal resection was performed en bloc with the bladder, prostate, seminal vesicles, urethra, ureter, uterus, ovaries or sacrum as indicated (Figure 2). When tumor was found to abut the prostate without infiltrating the parenchyma, bladder sparing partial prostatectomy was performed. Lateral pelvic lymph node dissection was performed when there were residual nodes after neoadjuvant therapy. A Wallace 66-type uretero-ileal conduit was performed for urinary reconstruction. Pelvic floor reconstruction was performed with omental flap, biological mesh, or musculofascial flaps depending on the requirement, availability, and the logistics.
All the patients who were fit to tolerate chemotherapy were given adjuvant chemotherapy, which was based on the final histopathological examination determining the response to initial NACTRT. After completion of treatment, each patient was followed up every 3 months for first 2 years after surgery, and every 6 months after that for next 3 years. At the time of follow-up, history, physical examination, and serum CEA levels were checked. CECT abdomen and colonoscopy were performed at 1year after the treatment completion.
Clinical outcomes that were assessed included median blood loss, need for plastic reconstruction, and 30-day postoperative morbidity and mortality. Short-term oncological outcomes that were assessed included CRM positivity, tumor site perforation (TSP), and distal margin involvement. Complete resection was defined as resection of all pelvic disease with a CRM of more than 1 mm. CRM of less than 1 mm was considered as residual disease. OS was defined as the time from the date of surgery to the date of last follow-up or death. Disease-free survival (DFS) was defined as the time from the date of surgery to the date of detection of recurrence. Recurrence was classified as local or distant. Local recurrence was defined as tumors that recurred in the pelvic cavity and the anastomotic area.
Statistical analysis
All data were entered and analyzed using SPSS, version 20.0 (SPSS, Chicago, IL, USA). Survival rate analysis was performed using a Kaplan-Meier survival curve. The following independent variables were included the univariate analysis of survival—metastatic status at presentation, NACTRT, pathological tumor stage (pT), pathological nodal stage (pN), CRM positivity, TSP, tumor regression grade (TRG), and adjuvant therapy. A P value of <0.05 was considered to be statistically significant.
Results
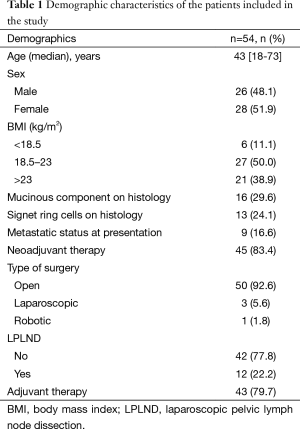
There were 54 MVRs performed between 1 July 2013 and 31 May 2015, and all were included in the analysis. The baseline patient characteristics are shown in Table 1. All but one patient had primary rectal adenocarcinoma. Nine patients (16.9%) did not receive neoadjuvant therapy—one patient had previously received pelvic irradiation, and rest eight patients showed tumor situated above the peritoneal reflection. Forty-five patients received neoadjuvant therapy (83.4%)—6 received neoadjuvant chemotherapy and 39 received NACTRT. Among those who received NACTRT, 13 patients (33%) received additional chemotherapy in view of threatened CRM on MRI 2.
Full table
Nine patients (16.6%) had distant metastasis at presentation—six with liver metastasis, one with metastasis in liver and para aortic nodes, one with isolated para aortic nodes and one with abdominal wall deposits with limited peritoneal disease. Among the seven patients with liver metastasis, one underwent surgical resection after completion of primary surgery and adjuvant therapy and one underwent radiofrequency ablation. Among the rest of the five patients, liver lesions disappeared completely after the neoadjuvant therapy, and, hence, patients were kept under surveillance. One patient with abdominal wall deposit and limited peritoneal disease underwent surgical resection of the metastatic deposits along with the primary surgery.
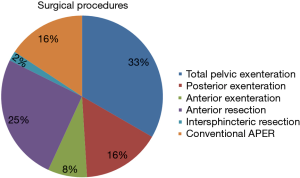
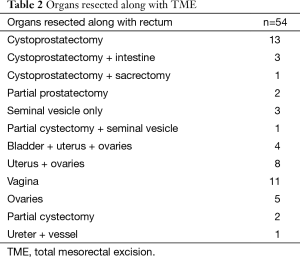
Details of the surgical procedures are shown in Figure 3. Eighteen patients (33%) underwent sphincter preservation with primary colorectal or coloanal anastomosis. The number and spectrum of organs resected are shown in Table 2. Median blood loss was 1,300 mL (range, 100–4,000 mL). Median blood replaced in peri operative period was 150 mL (range, 0–2,100 mL). Perineal reconstruction was done with musculofascial flaps in 10 patients (18.5%), which included gluteal flap in 8 and pedicled anterolateral thigh flap in 2. Biological mesh was used for reconstruction in additional four patients (7.4%). Median hospital stay was 12 days (range, 6–40 days).
Full table
There was one peri operative death (1.9%). This patient had undergone anterior exenteration and developed anastomotic leakage. He underwent exploratory laparotomy with disconnection of the anastomosis. Three weeks after the initial exploration, he developed leakage from ileal conduit. He was re-explored with construction of new ileal conduit. However, he showed stormy postoperative course and died due to septicemia. Postoperative morbidity (grade 2/3/4) developed in 20 patients (37%). Details of the complications are mentioned in Table 3.

Full table
Pathological characteristics of the patients are mentioned in Table 4. Complete response was seen in six patients (11.2%). Among the patients with sphincter preservation, median distal margin was 3.5 cm, and none revealed the distal margin involvement by the tumor. One patient showed proximal margin involvement by the tumor. This patient had undergone an abdominoperineal excision of the rectum, and grossly, the proximal margin was 6 cm but showed sub mucosal disease tracking upto 6 cm. He had an end sigmoid stoma, which was revised during the second surgical procedure. Among the six patients (10%) with involved CRM, two (33%) developed local recurrence and three developed distant recurrence (50%). All the patients with involved CRM received NACTRT with two of the six patients given additional chemotherapy after NACTRT.

Full table
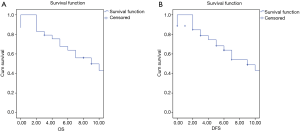
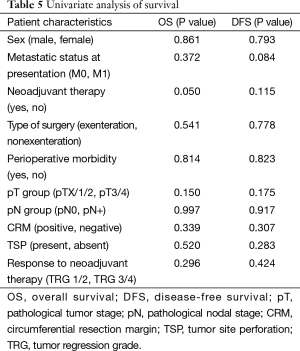
Median follow-up duration was 9 months (range: 3–22 months). One patient was lost to follow-up. Four patients developed local recurrence (7.4%), 10 developed distant recurrence (18.5%), and 40 were recurrence free at the last follow-up. Among the four patients with local recurrence, two showed positive CRM and one involved proximal margin. The fourth patient presented initially with tumor perforation with pyoperitoneum. She underwent exploratory laparotomy with proximal diversion loop ileostomy and was treated with palliative intent. However, she showed excellent response to chemotherapy and, hence, was offered MVR. All the four patients showed recurrence within first year after surgery and, hence, were treated with palliative chemotherapy. Among the 10 patients with distant recurrence, 2 patients each showed metastatic para-aortic nodes, extensive peritoneal deposits, bi-lobar liver metastasis and extensive multi organ metastasis, and one patient each showed abdominal wall deposits and extensive inguinal nodal deposits. Estimated 5-year OS was 70%; however, median OS and DFS were not reached (Figure 4). On univariate analysis, none of the factors significantly affected the survival or recurrence except for neoadjuvant therapy (Table 5).
Full table
Discussion
This is a retrospective review of a prospectively maintained database evaluating the outcomes for patients of LARCs treated by radical rectal resection en bloc with the adjacent involved organ. Although there are previous studies showing the role of MVR, most of these studies are being conducted in the western population. In these studies, the median age of the patients included is around 60 years (Table 6). In contrast to these studies, Indian patients are younger (median age in this study was 43 years). In addition, significant proportion of these patients showed signet ring cells (24.1%) and mucinous component (29.6%). In these aggressive subtypes with inferior oncological outcomes, impaired urinary and sexual functions resulting from the MVRs may not be acceptable to the younger patients. So, this study was undertaken to determine the feasibility and safety of the procedure in the Indian population.
Consistent with prior studies, this series noted a preponderance of female (51.9%) over male subjects in contrast to standard rectal resections (11,14). Proportion of women is higher because of the fact that low rectal cancers can more easily invade the vagina than the prostate, since the latter is covered by a dense capsule. The rate of sphincter preserving surgery was significantly lower in this series (33%), which is consistent with the results of other studies as well (11,14). The low rate of sphincter preservation in these surgeries is because of the relatively large size of the tumors, the technical difficulty of resection of a bulky tumor by a solely anterior approach and the reluctance of surgeons to perform an anastomosis below the peritoneal reflection in the absence of surrounding pelvic structures (15). Peri operative morbidity and mortality rates of 37% and 1.85%, respectively, noted in this series are comparable with the literature, which shows rates ranging from 18% to 77.8% and 0% to 13.3% for peri operative morbidity and mortality, respectively (16).
It is a common perception that involved CRM on MRI is a predictor of inferior DFS and OS. Although this is true in cases of standard total mesorectal excision (TME), MVR results in the removal of the involved viscera and, hence, should hypothetically lead to lower local recurrence. However, whether this transforms into improved overall outcomes is yet to be proven. Long-term results of the MERCURY study group have shown that, outcome of patient who revealed an involved CRM on MRI, which was confirmed on final histopathological examination, showed inferior outcomes (5-year DFS and OS of 21.6% and 25.1%, respectively) compared with those with involved CRM on MRI but clear CRM on final histopathological examination (5-year DFS and OS of 66% and 70%, respectively) (5). The other area of controversy is to determine the extent of resection based on MRI. Whether extent of resection should be based on MRI 1 or MRI 2 is debatable. In this study, the surgical decision on MVRs was based on MRI 2 in view of young age of the patients. Whether this will translate into similar oncological outcomes is difficult to determine. In spite of deciding the surgical management based on MRI 2, pathological involvement of the adjacent organs was seen in only 46% patients in this series.
Achieving complete resection has been shown to be the most important prognostic factor (11). The complete resection rate of 87% noted in this series is within the range for primary rectal carcinoma in contemporary series (10,13). In this series, among the seven patients with positive resection margins, most commonly involved margin was CRM (six patients). Overall, five patients with involved margins developed recurrences (local/distant) within first year of the primary surgery (85%) in spite of adjuvant therapy. On univariate analysis, CRM positivity did not significantly influence local recurrence or survival. This may reflect small patient numbers.
The use of neoadjuvant treatment to improve R0 resection rates and, hence, loco regional control and survival is the standard of care in the management of LARC (17,18). The role of neoadjuvant therapy in these aggressive malignancies has been highlighted by the following results in this series. Although all the tumors included in this series were clinical T4 at presentation, only 46.3% tumors were pT4. The patients with pT0–T2 showed better OS and DFS compared with pT3–T4 (although it did not reach statistical significance). In addition, when the survival was compared between those who received neoadjuvant therapy and those who underwent upfront surgery, it was found that neoadjuvant therapy significantly affected survival. The impact of down staging effect of neoadjuvant treatment on survival that is observed in this series is consistent with the other series (11,19). While most patients received NACTRT in this series, some were operated upfront as the need for MVRs was found intra-operatively. Some of these were the patients with upper rectal cancers who underwent resection of the involved small bowel. Currently, there is no level 1 evidence for the use of additional chemotherapy after NACTRT. In this series, among the 13 patients who received additional chemotherapy after NACTRT, 2 showed complete response, and only 5 revealed pT4 (37%). Although the numbers are too small to determine the impact of such a therapy on survival, this series do show improved R0 resection as a result of further down staging.
The role of extended lymphadenectomy in the management of rectal cancers is highly debated. In spite of extended TME performed in the majority of patients in this series, extended lymphadenectomy was offered only to those who showed residual nodal disease after NACTRT, which is consistent with the results of the other studies (20). For this reason, it is not possible to comment on the advantages of extended lymph node resection in these patients. However, node positivity did not influence survival or recurrence in this series. This is in contrast to few studies on pelvic exenteration, which found nodal status as an important prognostic factor influencing survival (21,22). However, a recent meta-analysis did show that there was no improvement in survival and local recurrences when an extended lymphadenectomy was performed compared with standard TME (23).
Recent systemic review on MVR showed that 5-year OS rates ranged from 33% to 74.1% and DFS from 25.9% to 52% (16). Considering the fact that 29.6% patients showed mucinous component on final histology and 24.1% patients showed signet ring cell adenocarcinoma, our short-term results seem to be comparable (Table 6). However, in young patients with biologically aggressive tumors such as mucinous or signet ring cell adenocarcinoma, long-term outcomes are difficult to predict, and, hence, longer follow-up is needed. Limitations of the present series include retrospective nature of the study, limited follow-up and fewer patients included in the study.

Full table
Conclusions
In Indian subcontinent, MVRs in young patients with high proportion of signet ring cell adenocarcinomas based on MRI 2 is associated with similar CRM involvement and similar adjacent organ involvement as the western patients who are older and surgery being planned on MRI 1. However, longer follow-up is needed to confirm noninferiority of oncological outcomes.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2015;136:E359-86. [Crossref] [PubMed]
- van Gijn W, Marijnen CA, Nagtegaal ID, et al. Preoperative radiotherapy combined with total mesorectal excision for resectable rectal cancer: 12-year follow-up of the multicentre, randomised controlled TME trial. Lancet Oncol 2011;12:575-82. [Crossref] [PubMed]
- Wibe A, Rendedal PR, Svensson E, et al. Prognostic significance of the circumferential resection margin following total mesorectal excision for rectal cancer. Br J Surg 2002;89:327-34. [Crossref] [PubMed]
- Bernstein TE, Endreseth BH, Romundstad P, et al. Circumferential resection margin as a prognostic factor in rectal cancer. Br J Surg 2009;96:1348-57. [Crossref] [PubMed]
- Taylor FG, Quirke P, Heald RJ, et al. Preoperative magnetic resonance imaging assessment of circumferential resection margin predicts disease-free survival and local recurrence: 5-year follow-up results of the MERCURY study. J Clin Oncol 2014;32:34-43. [Crossref] [PubMed]
- Eveno C, Lefevre JH, Svrcek M, et al. Oncologic results after multivisceral resection of clinical T4 tumors. Surgery 2014;156:669-75. [Crossref] [PubMed]
- Vermaas M, Ferenschild FT, Verhoef C, et al. Total pelvic exenteration for primary locally advanced and locally recurrent rectal cancer. Eur J Surg Oncol 2007;33:452-8. [Crossref] [PubMed]
- Wiig JN, Poulsen JP, Larsen S, et al. Total pelvic exenteration with preoperative irradiation for advanced primary and recurrent rectal cancer. Eur J Surg 2002;168:42-8. [Crossref] [PubMed]
- de Wilt JH, Vermaas M, Ferenschild FT, et al. Management of locally advanced primary and recurrent rectal cancer. Clin Colon Rectal Surg 2007;20:255-63. [Crossref] [PubMed]
- Harris DA, Davies M, Lucas MG, et al. Multivisceral resection for primary locally advanced rectal carcinoma. Br J Surg 2011;98:582-8. [Crossref] [PubMed]
- Smith JD, Nash GM, Weiser MR, et al. Multivisceral resections for rectal cancer. Br J Surg 2012;99:1137-43. [Crossref] [PubMed]
- Mañas MJ, Espín E, López-Cano M, et al. Multivisceral resection for locally advanced rectal cancer: prognostic factors influencing outcome. Scand J Surg 2014;104:154-60. [Crossref] [PubMed]
- Crawshaw BP, Augestad KM, Keller DS, et al. Multivisceral resection for advanced rectal cancer: outcomes and experience at a single institution. Am J Surg 2015;209:526-31. [Crossref] [PubMed]
- Mathis KL, Nelson H, Pemberton JH. Can unresectable colorectal cancer be cured? Adv Surg 2009;43:211-9. [Crossref] [PubMed]
- Park IJ, Yu CS. Current issues in locally advanced colorectal cancer treated by preoperative chemoradiotherapy. World J Gastroenterol 2014;20:2023-9. [Crossref] [PubMed]
- Mohan HM, Evans MD, Larkin JO, et al. Multivisceral resection in colorectal cancer: a systematic review. Ann Surg Oncol 2013;20:2929-36. [Crossref] [PubMed]
- Ratto C, Valentini V, Morganti AG, et al. Combined-modality therapy in locally advanced primary rectal cancer. Dis Colon Rectum 2003;46:59-67. [Crossref] [PubMed]
- Theodoropoulos G, Wise WE, Padmanabhan A, et al. T-level downstaging and complete pathologic response after preoperative chemoradiation for advanced rectal cancer result in decreased recurrence and improved disease-free survival. Dis Colon Rectum 2002;45:895-903. [Crossref] [PubMed]
- Kaminsky-Forrett MC, Conroy T, Luporsi E, et al. Prognostic implications of downstaging following preoperative radiation therapy for operable T3-T4 rectal cancer. Int J Radiat Oncol Biol Phys 1998;42:935-41. [Crossref] [PubMed]
- Akiyoshi T, Ueno M, Matsueda K, et al. Selective lateral pelvic lymph node dissection in patients with advanced low rectal cancer treated with preoperative chemoradiotherapy based on pretreatment imaging. Ann Surg Oncol 2014;21:189-96. [Crossref] [PubMed]
- Derici H, Unalp HR, Kamer E, et al. Multivisceral resections for locally advanced rectal cancer. Colorectal Dis 2008;10:453-9. [Crossref] [PubMed]
- Ike H, Shimada H, Yamaguchi S, et al. Outcome of total pelvic exenteration for primary rectal cancer. Dis Colon Rectum 2003;46:474-80. [Crossref] [PubMed]
- Georgiou P, Tan E, Gouvas N, et al. Extended lymphadenectomy versus conventional surgery for rectal cancer: a meta-analysis. Lancet Oncol 2009;10:1053-62. [Crossref] [PubMed]


