
Hematologic markers of distant metastases in gastric cancer
Introduction
Gastric cancer is the second most common gastrointestinal malignancy after colorectal cancer (CRC) with one million cases diagnosed worldwide annually (1). Around 40% of gastric cancer patients have distant metastases at the time of diagnosis (2). Despite the recent advancements in the management of metastatic gastric cancer, the prognosis remains poor with a 5-year survival rate of less than 4% (3).
Inflammation has been linked to carcinogenesis, metastases and survival outcomes in cancer (4,5). However, the direct relationship between inflammation and metastases is unclear. Absolute neutrophil count (ANC), absolute lymphocyte count (ALC), absolute eosinophil count (AEC), absolute monocyte count (AMC), monocyte-lymphocyte ratio (MLR), platelet-lymphocyte ratio (PLR) and neutrophil-lymphocyte ratio (NLR) were found to have a negative impact on survival in various tumors including gastric cancer (6-10). Additionally; recent studies showed that high NLR is an independent predictive factor for baseline presence of brain metastases in advanced non-small cell lung cancer (NSCLC), and for the baseline presence of lung metastases in advanced CRC (11,12).
Various explanations have been proposed to describe the relationship between inflammation and metastases. High neutrophil count resulting in high NLR is thought to promote metastases via releasing certain growth factors such as VEGF and other proteases (13). Alternatively, lymphocytes have an essential role in eliminating tumor cells by inducing cytotoxic cell death and impeding tumor cell proliferation and migration, explaining the association of low ALC with poor outcomes in cancer due to diminished immune response to malignancy (14,15).
In this retrospective study, we aim to further identify the relationship between blood inflammatory cells with distant metastases in gastric cancer as this might have important implications on the management of gastric cancer. Our proposed hypothetical questions were, do gastric cancer patients with high NLR have a high incidence of distant metastases? Do they need more frequent surveillance imaging? Do they benefit from prophylactic strategies? And finally, might they respond better to the immunotherapy?
Methods
This is a retrospective chart review study that was approved by the Institutional Review Board office at King Hussein Cancer Center (IRB Approval No. 18KHCC54). Informed consent is not needed since it is a retrospective chart review study. Five hundred and two patients diagnosed with gastric cancer, who received treatment at KHCC (Amman, Jordan), were included. Positron emission tomography (PET) scans, computed tomography (CT) scans and magnetic resonance imaging (MRI) were used to detect distant metastases. Clinical data including age, gender, location of primary tumor, and the presence of baseline distant metastases were collected and summarized in Table 1. Patients were excluded if they were on steroids before obtaining a complete blood count (CBC). Using steroids was expected to confound the results of the study as it leads to leukocytosis (16) and specifically neutrophilia (17).

Full table
CBC with differential white cell count at the time of diagnosis was collected before the initiation of any cancer specific treatment (systemic treatment or radiation). The pre-treatment baseline NLR, MLR and PLR were calculated using these formulas: NLR = ANC/ALC, MLR = AMC/ALC and PLR = platelet count/ALC.
The receiver operating characteristic (ROC) curve was used to determine the optimal cut-off value for NLR associated with distant metastases, matching the most extreme joint sensitivity and specificity. The association between NLR and clinical factors including the age, gender, and the location of primary tumor with the presence of distant metastases were examined. Univariate and multivariate logistic regression analyses were used to test the association between the various variables and presence of distant metastases on diagnosis. A P value of ≤0.05 was determined as the cut off value for a significant association.
Our analysis proceeded stepwise. In the first phase, we examined the association between baseline NLR with the presence of distant metastases. In the second phase, we examined the association between other hematologic parameters including ANC, ALC, AEC, AMC, MLR & PLR with the baseline presence of distant metastases. In the third phase, we examined the association between baseline presence of distant metastases with the clinical variables like age, gender, and location of the primary tumor. In the fourth phase, we performed a multivariate analysis that included the collected variables (age, gender, and location of primary tumor) with NLR as a continuous variable, to identify correlation with baseline distant metastases. In the last phase, the fifth, we examined the association between the hematologic parameters including ANC, ALC, AEC, AMC, NLR, MLR & PLR with overall survival (OS).
Results
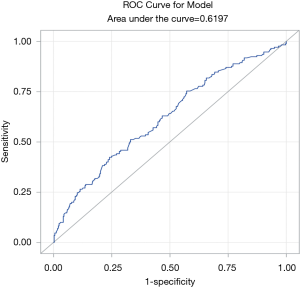
The clinical features of 502 gastric cancer patients are summarized in Table 1. More than half of the patients were males (56.0% versus 44.0%, with a male to female ratio of 1.3:1). The median age at diagnosis was 54 years old. The median OS for all patients was 12 months. The baseline NLR mean was 4.4, and the median was 3.3. The optimal NLR cutoff value to correlate with distant metastases was determined to be 3.9 using the ROC curve where the area under curve (AUC) was 0.6197 (Figure 1).
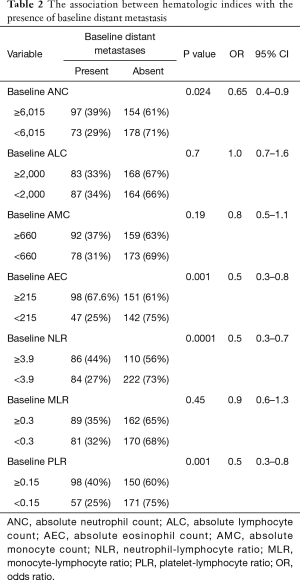
The relationship between the various hematologic cell counts, their ratios and the baseline presence of distant metastases are shown in Table 2. Patients with high baseline NLR (≥3.9) were more likely to have distant metastases at diagnosis in comparison to patients with low baseline NLR (<3.9) [P=0.0001, odds ratio (OR): 0.5, 95% CI: 0.3–0.7]. High baseline ANC (≥6,015/µL), high AEC (≥215 /µL), and high PLR (≥0.15) were significantly associated with baseline distant metastases (P=0.024, 0.001, and 0.001 respectively). The ROC curve was utilized to determine which parameter including NLR, ANC, AEC, or PLR is a better predictor of baseline presence of distant metastases (Figure 2). As shown, NLR has the highest AUC followed by ANC, PLR, and then AEC which indicates that NLR is a better predictor of distant metastases in comparison to the other tested parameters. Accordingly, NLR was the only parameter that was included in the subsequent multivariate analysis. ALC, AMC, and MLR did not show significant association with the presence of baseline distant metastases in gastric cancer (P=0.7, 0.19 and 0.45, respectively).
Full table
Table 3 shows the univariate and multivariate analyses assessing the association of NLR and clinical variables with baseline distant metastases. Age was significantly associated with the baseline presence of distant metastases. Younger patients (≤54 years) had more distant metastases compared to older ones (>54 years) (P=0.014). Gender and location of the primary tumor were not associated with baseline presence of distant metastases in the univariate analysis (P=0.32 and 0.33, respectively). Despite of that; gender and location of the primary tumor were included in the multivariate analysis because of their potential clinical relevance. In the multivariate analysis, NLR remained significantly associated with the baseline presence of distant metastases (P=0.0005).
Full table
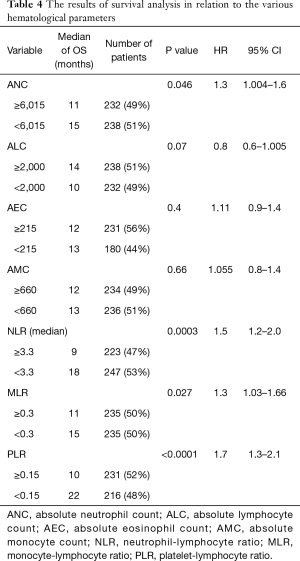
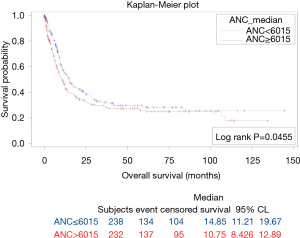
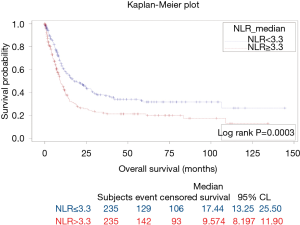
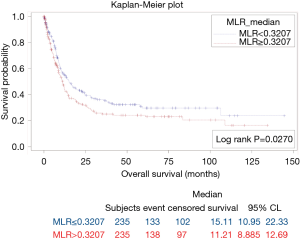
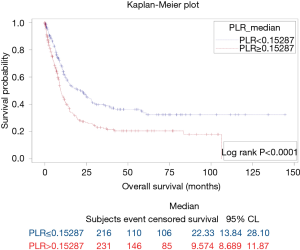
The results of survival analysis are summarized in Table 4. High baseline ANC, NLR, MLR, and PLR were associated with poor OS (P=0.0455, 0.0003, 0.0270, and <0.0001, respectively) (Figures 3-6). Baseline ALC, AEC, and AMC were not associated with the OS (P=0.07, 0.4, and 0.66, respectively).
Full table
Discussion
To our knowledge, this is the first study assessing the potential predictive value of NLR for detecting distant metastases in patients with gastric cancer. An elevated baseline NLR (≥3.9) was an independent factor associated with the baseline presence of distant metastases in gastric cancer as seen in univariate and multivariate analyses after adjusting for important covariates (P=0.0001, and P=0.0005, respectively) (Table 3). Additionally; patients with elevated baseline ANC, AEC, and PLR had more distant metastases in comparison to patients with low baseline ANC, AEC, and PLR (P=0.024, 0.001 and 0.001, respectively). Thus, basic hematological parameters might be of value in predicting the baseline presence of distant metastases in patients with gastric cancer.
The presence of distant metastases is the most worrisome prognostic feature of gastric cancer (18). Using simple tests such as CBC to predict the presence of distant metastasis upon diagnosis, and probably predicting the likelihood of subsequent development of distant metastases, might have a direct impact on gastric cancer patients’ care. Surveillance for distant metastases through more frequent imaging, and consideration for prophylactic approaches in patients with high NLR could be an attractive area for future research.
Neutrophil to lymphocyte ratio was shown to correlate with PD1/PD-L1 expression, and with response to chemotherapy and immunotherapy in various cancers (19-23). NLR was found to be associated with immune checkpoint molecules PD1\PD-L1 expression in gastric cancer and biliary tract cancer which likely indicates that NLR could be used as a marker for PDL-1 expression in solid tumors (24,25).
Treatment of distant metastases in gastric cancer includes radiation, chemotherapy, immunotherapy, and rarely surgery. Whether NLR is associated with response of the distant metastases to treatment is unknown. A major advancement in the management of gastric cancer will be achieved if NLR is confirmed to be a predictive marker of distant metastases’ response to immunotherapy or other available treatments.
Limitations of this study include the retrospective nature of the data with all cases collected from a single cancer center. There was no stratification by the number and sites of distant metastatic lesions. There was no assessment of the exact treatments delivered. Additionally, no solid data on the confounding factors that might elevate NLR such as infections were provided.
Conclusions
Elevated baseline NLR is an independent predictive factor for the presence of baseline distant metastases in gastric cancer. This inflammatory-distant metastases association is worthy of further studying in prospective trials, and might be used as a stratification tool to assess response of distant metastases to systemic treatments such as immunotherapy. This might also have implications regarding distant metastases surveillance in patients with high NLR, and probably considering prophylactic approaches in this patient subset.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the Institutional Review Board Office at King Hussein Cancer Center (IRB Approval No: 18KHCC54).
References
- Ferlay J, Soerjomataram I, Dikshit R, et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2015;136:E359-86. [Crossref] [PubMed]
- Riihimäki M, Hemminki A, Sundquist K, et al. Metastatic spread in patients with gastric cancer. Oncotarget 2016;7:52307-16. [Crossref] [PubMed]
- Brenner H, Rothenbacher D, Arndt V. Epidemiology of stomach cancer. Methods Mol Biol 2009;472:467-77. [Crossref] [PubMed]
- Moore MM, Chua W, Charles KA, et al. Inflammation and cancer: causes and consequences. Clin Pharmacol Ther 2010;87:504-8. [Crossref] [PubMed]
- Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell 2010;140:883-99. [Crossref] [PubMed]
- Xie D, Allen MS, Marks R, et al. Nomogram prediction of overall survival for patients with non-small-cell lung cancer incorporating pretreatment peripheral blood markers. Eur J Cardiothorac Surg 2018;53:1214-22. [Crossref] [PubMed]
- Zhang X, Zhang W, Feng LJ. Prognostic significance of neutrophil lymphocyte ratio in patients with gastric cancer: a meta-analysis. PLoS One 2014;9:e111906. [Crossref] [PubMed]
- Dirican A, Kucukzeybek BB, Alacacioglu A, et al. Do the derived neutrophil to lymphocyte ratio and the neutrophil to lymphocyte ratio predict prognosis in breast cancer? Int J Clin Oncol 2015;20:70-81. [Crossref] [PubMed]
- Minardi D, Scartozzi M, Montesi L, et al. Neutrophil-to-lymphocyte ratio may be associated with the outcome in patients with prostate cancer. Springerplus 2015;4:255. [Crossref] [PubMed]
- Stotz M, Gerger A, Eisner F, et al. Increased neutrophil-lymphocyte ratio is a poor prognostic factor in patients with primary operable and inoperable pancreatic cancer. Br J Cancer 2013;109:416-21. [Crossref] [PubMed]
- Koh YW, Choi JH, Ahn MS, et al. Baseline neutrophil-lymphocyte ratio is associated with baseline and subsequent presence of brain metastases in advanced non-small-cell lung cancer. Sci Rep 2016;6:38585. [Crossref] [PubMed]
- Abu-Shawer M, Abu-Shawer O, Souleiman M, et al. Hematologic Markers of Lung Metastasis in Stage IV Colorectal Cancer. J Gastrointest Cancer 2018. [Epub ahead of print]. [Crossref] [PubMed]
- Swierczak A, Mouchemore KA, Hamilton JA, et al. Neutrophils: important contributors to tumor progression and metastasis. Cancer Metastasis Rev 2015;34:735-51. [Crossref] [PubMed]
- Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity 2013;39:1-10. [Crossref] [PubMed]
- Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet 2001;357:539-45. [Crossref] [PubMed]
- Shoenfeld Y, Gurewich Y, Gallant LA, et al. Prednisone-induced leukocytosis. Influence of dosage, method and duration of administration on the degree of leukocytosis. Am J Med 1981;71:773-8. [Crossref] [PubMed]
- Nakagawa M, Terashima T, D'yachkova Y, et al. Glucocorticoid-induced granulocytosis: contribution of marrow release and demargination of intravascular granulocytes. Circulation 1998;98:2307-13. [Crossref] [PubMed]
- Yang D, Hendifar A, Lenz C, et al. Survival of metastatic gastric cancer: Significance of age, sex and race/ethnicity. J Gastrointest Oncol 2011;2:77-84. [PubMed]
- Tanaka H, Tamura T, Toyokawa T, et al. Clinical Relevance of Postoperative Neutrophil-Lymphocyte Ratio (NLR) to Recurrence After Adjuvant Chemotherapy of S-1 for Gastric Cancer. Anticancer Res 2018;38:3745-51. [Crossref] [PubMed]
- Templeton AJ, Knox JJ, Lin X, et al. Change in Neutrophil-to-lymphocyte Ratio in Response to Targeted Therapy for Metastatic Renal Cell Carcinoma as a Prognosticator and Biomarker of Efficacy. Eur Urol 2016;70:358-64. [Crossref] [PubMed]
- Liu D, Jin J, Zhang L, et al. The Neutrophil to Lymphocyte Ratio May Predict Benefit from Chemotherapy in Lung Cancer. Cell Physiol Biochem 2018;46:1595-605. [Crossref] [PubMed]
- Moschetta M, Uccello M, Kasenda B, et al. Dynamics of Neutrophils-to-Lymphocyte Ratio Predict Outcomes of PD-1/PD-L1 Blockade. Biomed Res Int 2017;2017:1506824. [Crossref] [PubMed]
- Khunger M, Patil PD, Khunger A, et al. Post-treatment changes in hematological parameters predict response to nivolumab monotherapy in non-small cell lung cancer patients. PLoS One 2018;13:e0197743. [Crossref] [PubMed]
- Sangkhamanon S, Jongpairat P, Sookprasert A, et al. Programmed Death-Ligand 1 (PD-L1) Expression Associated with a High Neutrophil/Lymphocyte Ratio in Cholangiocarcinoma. Asian Pac J Cancer Prev 2017;18:1671-4. [PubMed]
- Tanaka H, Tamura T, Kimura K, et al. Association of the immune checkpoint molecule expression with neutrophil-lymphocyte ratio in patients with gastric cancer: a retrospective study. J Clin Oncol 2016;34:48. [Crossref]


