A margin distance analysis of the impact of adjuvant chemoradiation on survival after pancreatoduodenectomy for pancreatic adenocarcinoma
Introduction
Surgical resection represents the only potential curative modality in the treatment of pancreatic ductal adenocarcinoma (PDA) (1-3). The 5-year survival following surgical extirpation remains around 15–20%, therefore efforts to identify the optimal adjuvant regimen remain a priority. Although adjuvant chemotherapy has been shown to improve survival in several phase 3 trials, the role of adjuvant chemoradiotherapy (CRT) remains controversial (2,4-7). Data from available studies are confounded by heterogeneity in the dose and timing of adjuvant radiotherapy (RT), as some trials lacked quality control, utilized ineffective RT doses, or employed split-course RT that mandated treatment interruption (2,8,9). Additionally, adjuvant therapy trials prior to ESPAC-1 compared CRT to observation alone, utilized suboptimal chemotherapy, included patients with other periampullary malignancies, and lacked large sample sizes to allow for subgroup analysis to determine which patients derived benefit from systemic chemotherapy versus CRT.
The above-mentioned studies also had variability in the definition of R0 and R1 margins, with most defining R0 as the absence of tumor cells at the cut margins regardless of margin distance (10,11). Standardized protocols of margin assessment for pancreaticoduodenectomy (PD) specimens, coupled with observations that increased margin clearance (MC: distance in mm between closest margin and tumor) is associated with improved survival, have suggested that R0 be defined as the absence of tumor cells within 1mm of any of the cut margins (12-17). Despite this, a consensus definition of what constitutes a margin-negative resection is lacking.
Based on the above limitations, the role of adjuvant CRT in patients with known MC remains undefined. This study sought to analyze the impact of adjuvant CRT on survival for PDA based on differential MC distances following PD. We hypothesized that patients with close margins would derive the greatest benefit from adjuvant CRT.
Methods
Case selection
After obtaining Institutional Review Board approval, a retrospective review of all patients who underwent PD at the University of Pittsburgh Medical Center between January 1, 2002 and December 31, 2014 was performed. Patients were excluded from analysis if the pathology report did not specify MC distance, if they received neoadjuvant RT, adjuvant RT alone, had a gross macroscopic (R2) positive resection, or if they suffered mortality within 90 days of their operative date.
Patient variables and definitions
Charts were abstracted for demographics, treatment, pathologic variables, recurrence, and death. Patient variables included age, sex, and body mass index (BMI). Treatment variables included receipt of neoadjuvant and/or adjuvant chemotherapy (defined as receipt of ≥1 treatment of single or multi-agent regimen), concomitant superior mesenteric vein/portal vein resection at the time of PD, and receipt, type (stereotactic vs. external beam), and dose of adjuvant RT (administered within 6 months of surgery and prior to a documented recurrence). External beam radiotherapy (EBRT) was delivered with either a 3-dimensional conformal or intensity-modulated technique with institutional clinical pathway recommendations for treatment of the resection bed and regional lymph node basins. Stereotactic beam radiotherapy (SBRT) was delivered via either CyberKnife (Accurray, Sunnvale, CA, USA) or Trilogy/TrueBeam (Varian Medical Systems, Palo Alto, CA, USA) radiosurgical delivery platforms as previously described (18,19). Pathologic variables included tumor size, lymphovascular invasion (LVI), perineural invasion (PNI), and lymph node metastases. Margins evaluated were the pancreatic neck, common bile duct, vascular groove (SMV/PV), retroperitoneum/uncinate/SMA, and “other” (gastric and jejunal). MC was defined as the closest single margin distance (regardless of its location) from the tumor and was categorized as 0mm (tumor cells at the cut margin), ≤1 mm (tumor cells within 1 mm but not at the cut margin), or >1 mm (tumor cells more than 1 mm from the cut margin).
The decision to pursue adjuvant RT post-resection for was made after multidisciplinary consultation. Patients were typically seen at 3–6 months intervals postoperatively by the operating surgeon and/or treating medical oncologist with cross-sectional imaging. Recurrence-free survival (RFS) was calculated from the date of surgery to the date of first recurrence or last censored visit. Overall survival (OS) was defined from the date of diagnosis to the date of death or last censored visit.
Statistics
IBM SPSS 24.0 software for Macintosh was used for all data analysis (IBM, Armonk, NY, USA). Variables were assessed for normal distribution using the Shapiro-Wilk test. Continuous variables with normal distribution were summarized using mean and standard deviation. Differences between groups were analyzed using the independent samples t-test or Mann-Whitney U test as appropriate. Data not normally distributed were summarized using median and interquartile range (IQR). Differences between groups were analyzed using the nonparametric Wilcoxon rank-sum test or Kruskal-Wallis test as appropriate. Categorical data were compared using the Pearson Chi-squared test or Fisher’s exact test as appropriate. Multivariate analysis was performed by Cox proportional hazards on variables that were significant on univariate analysis (P<0.10). Median RFS and OS were determined using the Kaplan-Meier method and Log-Rank test. A P value of <0.05 was considered statistically significant.
Results
Overall cohort characteristics
A total of 310 patients were analyzed. Demographics and treatment related variables are summarized in Table 1. Median age was 70 years and 56% were males. Vascular resections were performed in 20% of the cohort. The SMV/PV groove (45%), retroperitoneum/uncinate (37%), and pancreatic neck (14%) comprised the majority of closest measured margins. A total of 62 patients received 5-FU- or gemcitabine-based adjuvant CRT (20%), of which 39 patients received SBRT and 23 patients received EBRT. Median dose for SBRT was 36 Gy in 3 fractions and median dose for EBRT was 50.4 Gy in 28 fractions. A total of 181 patients (58%) received either 5-FU- or gemcitabine-based adjuvant chemotherapy alone, whereas 67 patients (22%) received no adjuvant therapy at all. Median follow-up for surviving patients was 64 months. For the entire cohort, median RFS was 14 months and median OS was 24 months.

Full table
Increased MC is associated with improved RFS and OS
A total of 67 patients had a MC of 0 mm, 113 patients had a MC of ≤1 mm, and 130 had a MC of >1 mm. Progressive increase in MC was associated with improvement in RFS and OS. Median RFS was 8.8 months for patients with 0 mm margins, 13.3 months for patients with ≤1 mm margins, and 21.5 months for patients with >1 mm margins (P<0.01 for all pairs; Figure 1A). Median OS was 15.4 months with 0 mm margins, 22.6 months with ≤1 mm margins, and 36.9 months for >1 mm margins (P<0.02 for all pairs; Figure 1B).
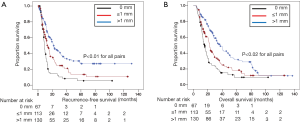
Clinicopathologic and treatment determinants of survival in the overall cohort
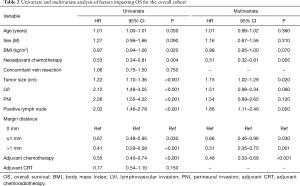
On univariate analysis, factors associated with increased risk of death in the overall cohort were increased age, increased tumor size, and the presence of LVI, PNI, or lymph node metastases (Table 2). Factors associated with reduced risk of death were increased BMI, neoadjuvant chemotherapy, increased MC (compared to 0 mm margins), and adjuvant chemotherapy. Adjuvant CRT was not associated with improved OS in the patient cohort as a whole (HR 0.77, 95% CI 0.54–1.10, P=0.15).
Full table
On multivariate analysis, increased MC was independently associated with improved OS (≤1 mm, HR 0.66; P=0.03; >1 mm, HR 0.51; P=0.001; compared to 0 mm). Additionally, neoadjuvant (HR 0.51, 95% CI 0.32–0.81, P=0.005) and adjuvant chemotherapy (HR 0.48, 95% CI 0.33–0.69, P<0.001) were also associated with improved OS, whereas increased tumor size (HR 1.15, 95% CI 1.02–1.29, P=0.02) and presence of lymph node metastases (HR 1.66, 95% CI 1.11–2.49, P=0.02) were independently associated with decreased OS.
Analysis of the impact of CRT on survival in the 0 mm and >1 mm margin subgroups
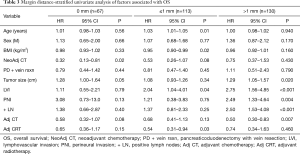
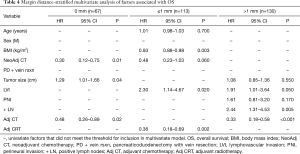
To delineate the association between adjuvant CRT and survival on MC, a margin-stratified univariate (Table 3) and multivariate (Table 4) analysis of clinic-pathologic and treatment-related factors was performed. In the 0 mm group (n=67), neoadjuvant chemotherapy (HR 0.30, 95% CI 0.12–0.75, P = 0.01), adjuvant chemotherapy (HR 0.48, 95% CI 0.26–0.89, P=0.02) and tumor size (HR 1.29, 95% CI 1.01–1.66, P=0.04) were independent predictors of OS (Table 4). In the >1 mm group (n=130), LVI (HR 1.91, 95% CI 1.01–3.64, P=0.05), lymph node metastases (HR 2.44, 95% CI 1.31–4.53, P=0.005) and adjuvant chemotherapy (HR 0.33, 95% CI 0.18–0.58, P<0.001) were independent predictors of OS. Adjuvant CRT was not a predictor of survival in the 0 mm and >1 mm margin groups.
Full table
Full table
Analysis of the impact of CRT on survival in the “close” margin (≤1 mm) subgroup
In patients with ≤1 mm margins (n=113), univariate analysis demonstrated age, BMI, presence of LVI and adjuvant CRT to be associated with survival (Table 3). On multivariate analysis, BMI (HR 0.93, 95% CI 0.88–0.98, P=0.003), LVI (HR 2.30, 95% CI 1.14–4.67, P=0.02), and adjuvant CRT remained as the only independent predictors of OS (Table 4). Specifically, adjuvant CRT was associated with a 64% reduction in mortality (HR 0.36, 95% CI 0.18–0.69, P=0.002).
Discussion
This study examined the association between adjuvant CRT and pancreatic cancer survival based on differential MC distances after PD. After first confirming that increased MC was associated with improved survival, adjuvant CRT was found to be associated with improved survival in the subset of patients with ≤1 mm MC but not the 0 mm or >1 mm cohort, suggesting that only a small subset of patients may derive clinical benefit from adjuvant CRT.
Prior studies that sought to evaluate the impact of adjuvant CRT following pancreatectomy for PDA demonstrate a spectrum of outcomes ranging from modest improvements to detrimental effects on survival (2,5,8,9). The lack of a standard definition of a margin-negative resection, amongst other factors, makes those results difficult to interpret. The GITSG trial demonstrated improved OS with the addition of adjuvant 5-FU and split-course RT compared to surgery alone (20 vs. 11 months); however, the study was small and did not meet accrual (8). Definition of margin status (R0 vs. R1) was not clearly outlined. EORTC 40891 failed to show a benefit with adjuvant 5-FU and RT compared to surgery alone (9,20). Similarly, margin definitions were unclear, patients with T3 tumors were excluded, and patients with ampullary adenocarcinoma and distal cholangiocarcinoma were included. In ESPAC-1, patients who received CRT with bolus 5-FU and 40 Gy of RT had decreased survival compared to those receiving chemotherapy alone (2). However, this trial has been criticized for its complex design and lack of RT quality control. Finally, two large meta-analyses of randomized trials demonstrated a benefit for adjuvant chemotherapy but no benefit for adjuvant CRT compared to surgery for PDA (21,22).
Since the above trials were limited by flawed design, small sample sizes, and poor quality control, a number of large retrospective series have attempted to evaluate the association between adjuvant CRT and outcomes (23-25). Herman et al. analyzed 908 patients at Johns Hopkins between 1993 and 2005 (23). Adjuvant 5-FU-based CRT was associated with improved median OS than those that underwent surgery alone (21.2 vs. 14.4 months). Subgroup analysis demonstrated adjuvant therapy was associated with improved survival in both R0 (24.3 vs. 17.0 months) and R1 (18.3 vs. 11.4 months) margins compared to observation. Corsini et al. reviewed 472 consecutive patients at the Mayo Clinic between 1975 and 2005 (24). Only patients who underwent R0 resection were included, and the addition of chemotherapy or CRT improved survival compared to observation (25.2 vs. 19.2 months). Hsu et al. analyzed the combined Johns Hopkins/Mayo Clinic experience and found similar results to each of the single institution reviews (25). Sugawara et al. performed a propensity-matched analysis of 2,532 patients from the Surveillance, Epidemiology, and End Results (SEER) database, and found adjuvant RT to be associated with improved survival compared to no RT (26). Finally, Kooby et al. analyzed 7288 patients from the National Cancer Database (NCDB) and determined that CRT, but not chemotherapy alone, was superior to no adjuvant therapy (27). This finding persisted on propensity-matched multivariate analysis.
Most of the above-cited prospective and retrospective reports either do not differentiate R0 from R1 margins, or do not provide data on MC. In contrast, this study analyzed the association between adjuvant CRT and survival based on margin distance and not on a “predefined” classification of R0 margins. As reported in earlier prospective trials, we found no association between CRT and survival of the overall patient cohort. However, we found CRT was associated with improved survival when restricted to a small group of “close margin” patients (MC ≤1 mm). The beneficial effect of CRT on close margins may be related to treatment of a smaller residual tumor burden compared to a larger one in the 0mm group. Conversely, the lack of benefit in the >1 mm group corroborates recent reports that a MC of >1 mm likely represents a true R0 resection, and that CRT may not be beneficial in this subgroup due to minimal or no residual localized disease. Future trials should account for margin distance when assessing the impact of CRT after PD for PDA.
Interestingly, despite the beneficial impact of CRT in the close margin subgroup, there was no difference in the local recurrence rate between patients who received adjuvant CRT and those who did not (30% vs. 23%; P=0.46) within this cohort. We did observe a decrease in distant recurrence in patients with ≤1 mm margins who received adjuvant CRT (37% vs. 59%; P=0.04). This finding could be explained by prior observations that local recurrence after PD is difficult to detect on cross sectional imaging (28). Autopsy series focusing on patterns of recurrence suggest that isolated distant failure is uncommon, and up to 70–80% of patients with distant recurrence also have local recurrence; however at least 15–20% of these local failures are undetectable by CT imaging (28,29). We obtained cross-sectional imaging every 3–6 months following PD. More frequent imaging could potentially allow for earlier detection of isolated local recurrence; however this strategy is not cost effective and has not been shown to improve survival in pancreatic cancer (30).
This analysis has several limitations. The study was retrospective in nature and suffers from the selection bias of including only PDs with reported margin distances. Additionally, although standardized protocols for PD specimen margin assessment have been shown to improve quality control and correlate with clinical outcomes, they were not uniformly utilized throughout this study since the study period spanned an earlier era when such protocols were largely unavailable. Although some patients were treated with SBRT on various protocols at our institution, the decision to pursue adjuvant CRT was largely made on a case-by-case basis by the surgeon and multidisciplinary team. A small sample size limited the ability to perform a separate analysis of the impact of the two RT modalities (SBRT vs. EBRT) on survival. Finally, it is possible that the benefit incurred by CRT in the ≤1 mm margin group may have been related to favorable disease characteristics in this cohort. Although patients with ≤1 mm margins who received CRT were younger, no other clinicopathologic factors were significantly different (Table S1). Furthermore, the multivariate analysis controlled for age and revealed CRT to be independently associated with improved OS in patients with ≤1 mm margins.

Full table
In conclusion, this study suggests that adjuvant CRT may be beneficial in the subset of patients who have a ≤1 mm MC after PD for PDA. This study underscores the importance of accurately assessing MC following PD, since this information can be used to further guide appropriate adjuvant treatment.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by Institutional Review Board (PRO16040384) and informed consent was taken from all the patients.
References
- Siegel RL, Miller KD, Jemal A. Cancer statistics, 2015. CA Cancer J Clin 2015;65:5-29. [Crossref] [PubMed]
- Neoptolemos JP, Stocken DD, Friess H, et al. A randomized trial of chemoradiotherapy and chemotherapy after resection of pancreatic cancer. N Engl J Med 2004;350:1200-10. [Crossref] [PubMed]
- Sener SF, Fremgen A, Menck HR, et al. Pancreatic cancer: a report of treatment and survival trends for 100,313 patients diagnosed from 1985-1995, using the National Cancer Database. J Am Coll Surg 1999;189:1-7. [Crossref] [PubMed]
- Neoptolemos JP, Stocken DD, Bassi C, et al. Adjuvant chemotherapy with fluorouracil plus folinic acid vs gemcitabine following pancreatic cancer resection: a randomized controlled trial. JAMA 2010;304:1073-81. [Crossref] [PubMed]
- Neoptolemos JP, Stocken DD, Dunn JA, et al. Influence of resection margins on survival for patients with pancreatic cancer treated by adjuvant chemoradiation and/or chemotherapy in the ESPAC-1 randomized controlled trial. Ann Surg 2001;234:758-68. [Crossref] [PubMed]
- Oettle H, Neuhaus P, Hochhaus A, et al. Adjuvant chemotherapy with gemcitabine and long-term outcomes among patients with resected pancreatic cancer: the CONKO-001 randomized trial. JAMA 2013;310:1473-81. [Crossref] [PubMed]
- Oettle H, Post S, Neuhaus P, et al. Adjuvant chemotherapy with gemcitabine vs observation in patients undergoing curative-intent resection of pancreatic cancer: a randomized controlled trial. JAMA 2007;297:267-77. [Crossref] [PubMed]
- Kalser MH, Ellenberg SS. Pancreatic cancer. Adjuvant combined radiation and chemotherapy following curative resection. Arch Surg 1985;120:899-903. [Crossref] [PubMed]
- Klinkenbijl JH, Jeekel J, Sahmoud T, et al. Adjuvant radiotherapy and 5-fluorouracil after curative resection of cancer of the pancreas and periampullary region: phase III trial of the EORTC gastrointestinal tract cancer cooperative group. Ann Surg 1999;230:776-82; discussion 82-4. [Crossref] [PubMed]
- Raut CP, Tseng JF, Sun CC, et al. Impact of resection status on pattern of failure and survival after pancreaticoduodenectomy for pancreatic adenocarcinoma. Ann Surg 2007;246:52-60. [Crossref] [PubMed]
- Albores-Saavedra J, Heffess C, Hruban RH, et al. Recommendations for the reporting of pancreatic specimens containing malignant tumors. The Association of Directors of Anatomic and Surgical Pathology. Am J Clin Pathol 1999;111:304-7. [Crossref] [PubMed]
- Chang DK, Johns AL, Merrett ND, et al. Margin clearance and outcome in resected pancreatic cancer. J Clin Oncol 2009;27:2855-62. [Crossref] [PubMed]
- Jamieson NB, Chan NI, Foulis AK, et al. The prognostic influence of resection margin clearance following pancreaticoduodenectomy for pancreatic ductal adenocarcinoma. J Gastrointest Surg 2013;17:511-21. [Crossref] [PubMed]
- Konstantinidis IT, Warshaw AL, Allen JN, et al. Pancreatic ductal adenocarcinoma: is there a survival difference for R1 resections versus locally advanced unresectable tumors? What is a "true" R0 resection? Ann Surg 2013;257:731-6. [Crossref] [PubMed]
- Esposito I, Kleeff J, Bergmann F, et al. Most pancreatic cancer resections are R1 resections. Ann Surg Oncol 2008;15:1651-60. [Crossref] [PubMed]
- Verbeke CS. Resection margins in pancreatic cancer. Surg Clin North Am 2013;93:647-62. [Crossref] [PubMed]
- Verbeke CS, Leitch D, Menon KV, et al. Redefining the R1 resection in pancreatic cancer. Br J Surg 2006;93:1232-7. [Crossref] [PubMed]
- Rwigema JC, Heron DE, Ferris RL, et al. Fractionated stereotactic body radiation therapy in the treatment of previously-irradiated recurrent head and neck carcinoma: updated report of the University of Pittsburgh experience. Am J Clin Oncol 2010;33:286-93. [PubMed]
- Rwigema JC, Heron DE, Parikh SD, et al. Adjuvant stereotactic body radiotherapy for resected pancreatic adenocarcinoma with close or positive margins. J Gastrointest Cancer 2012;43:70-6. [Crossref] [PubMed]
- Smeenk HG, van Eijck CH, Hop WC, et al. Long-term survival and metastatic pattern of pancreatic and periampullary cancer after adjuvant chemoradiation or observation: long-term results of EORTC trial 40891. Ann Surg 2007;246:734-40. [Crossref] [PubMed]
- Liao WC, Chien KL, Lin YL, et al. Adjuvant treatments for resected pancreatic adenocarcinoma: a systematic review and network meta-analysis. Lancet Oncol 2013;14:1095-103. [Crossref] [PubMed]
- Ren F, Xu YC, Wang HX, et al. Adjuvant chemotherapy, with or without postoperative radiotherapy, for resectable advanced pancreatic adenocarcinoma: continue or stop? Pancreatology 2012;12:162-9. [Crossref] [PubMed]
- Herman JM, Swartz MJ, Hsu CC, et al. Analysis of fluorouracil-based adjuvant chemotherapy and radiation after pancreaticoduodenectomy for ductal adenocarcinoma of the pancreas: results of a large, prospectively collected database at the Johns Hopkins Hospital. J Clin Oncol 2008;26:3503-10. [Crossref] [PubMed]
- Corsini MM, Miller RC, Haddock MG, et al. Adjuvant radiotherapy and chemotherapy for pancreatic carcinoma: the Mayo Clinic experience (1975-2005). J Clin Oncol 2008;26:3511-6. [Crossref] [PubMed]
- Hsu CC, Herman JM, Corsini MM, et al. Adjuvant chemoradiation for pancreatic adenocarcinoma: the Johns Hopkins Hospital-Mayo Clinic collaborative study. Ann Surg Oncol 2010;17:981-90. [Crossref] [PubMed]
- Sugawara A, Kunieda E. Effect of adjuvant radiotherapy on survival in resected pancreatic cancer: a propensity score surveillance, epidemiology, and end results database analysis. J Surg Oncol 2014;110:960-6. [Crossref] [PubMed]
- Kooby DA, Gillespie TW, Liu Y, et al. Impact of adjuvant radiotherapy on survival after pancreatic cancer resection: an appraisal of data from the national cancer data base. Ann Surg Oncol 2013;20:3634-42. [Crossref] [PubMed]
- Hishinuma S, Ogata Y, Tomikawa M, et al. Patterns of recurrence after curative resection of pancreatic cancer, based on autopsy findings. J Gastrointest Surg 2006;10:511-8. [Crossref] [PubMed]
- Iacobuzio-Donahue CA, Fu B, Yachida S, et al. DPC4 gene status of the primary carcinoma correlates with patterns of failure in patients with pancreatic cancer. J Clin Oncol 2009;27:1806-13. [Crossref] [PubMed]
- Tzeng CW, Abbott DE, Cantor SB, et al. Frequency and intensity of postoperative surveillance after curative treatment of pancreatic cancer: a cost-effectiveness analysis. Ann Surg Oncol 2013;20:2197-203. [Crossref] [PubMed]


