The utility of positron emission tomography/computed tomography in target delineation for stereotactic body radiotherapy for liver metastasis from primary gastric cancer: an illustrative case report and literature review
Introduction
The liver is a common site for metastatic disease for many cancers. Secondary liver metastases make up 95% of hepatic malignancies. Breast, lung, and gastrointestinal malignancies are often frequent sources of metastatic disease to the liver (1). Often, a single liver metastasis may be the only known focus of disease. Management of liver metastases may include surgical resection, systemic chemotherapy, chemoembolization, radioembolization, or external beam radiotherapy.
For patients unable to undergo surgery, radiotherapy offers a means of providing local disease control. Radiation therapy for liver metastases has evolved from whole liver radiotherapy to stereotactic body radiotherapy (SBRT), which involves the use of ablative radiation doses in fewer fractions. Reports on the use of SBRT have shown favorable rates of local disease control with few severe side effects (2).
In order to deliver SBRT safely and effectively, high-quality imaging is important for both radiation planning and radiation delivery. We report one case that highlights the importance of multiple imaging modalities in the target delineation phase of stereotactic radiation treatment of liver metastases.
Case presentation
The patient is a 75-year-old gentleman who was diagnosed in July 2013 with gastric adenocarcinoma after experiencing melena. He received neoadjuvant chemotherapy with docetaxel, 5-fluorouracil, and oxaliplatin followed by partial gastrectomy. He was followed with surveillance imaging until January 2015, at which point he was found to have a liver metastasis seen on FDG-PET imaging. The patient underwent surgical resection of the metastasis in February 2015. Postoperative imaging confirmed no residual disease. He received chemotherapy with paclitaxel and carboplatin. However, the patient could not tolerate this very well, so he was switched to ramucirumab, which he continued. In March 2016, CT scan of his chest, abdomen, and pelvis revealed a 1.7-cm hypodense lesion in segment 7 of the right lobe of the liver consistent with metastatic disease. He was determined not to be a candidate for further surgery on his liver. He was referred to the radiation oncology department at our institution for potential SBRT for this lesion.
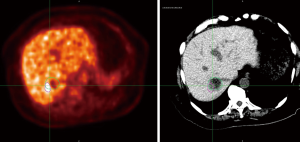
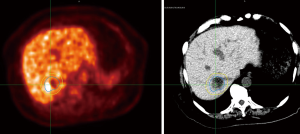
The patient was placed in a customized body mold and underwent four-dimensional (4D) CT simulation using a fast helical CT scanner while breathing freely. Images were sorted based on the patient’s respiratory cycle. An abdominal compression belt was used to decrease respiratory/diaphragmatic motion of the liver metastasis. As part of the patient’s radiation treatment planning, he underwent a limited FDG-PET scan of the liver while in the radiation treatment position with the customized body mold. The positron emission tomography/computed tomography (PET/CT) scan showed an area of the disease that extended outside of the area seen on CT imaging. Conversely, the simulation CT scan showed disease that was not fully characterized by the PET/CT scan. This can be seen in Figure 1. Information from both sets of images was used to define the radiation target volume to maximize the opportunity for disease control. A planning margin of 5 mm was used based on the motion of the tumor on 4D CT imaging. The patient ultimately received 45 Gy in 5 fractions to the PTV (Figure 2) using helical tomotherapy. Mega voltage CT imaging was obtained prior to each treatment session to verify radiation delivery to the planning target. Radiation therapy was delivered every other day with minimal acute toxicity.
Discussion
Image guidance is an important component for successful delivery of SBRT. In order to deliver ablative doses without significant toxicity to normal tissues and structures, the target must be outlined clearly and planning margins must be accurate. Typically, image-guidance for SBRT for liver disease has focused on movement of the target associated with the patient’s respiratory/diaphragmatic cycle. These image-guided techniques have included gated delivery of radiotherapy (e.g., breath-hold delivery or active breathing control) or tumor tracking (“tumor chasing”) using internal or external fiducial markers in combination with real-time imaging (orthogonal X-rays or fluoroscopy) (3).
However, target delineation is an important part of image-guided radiotherapy (IGRT). Multi-modality imaging techniques including MRI and PET/CT have been used for SBRT of liver metastases (3,4). PET/CT imaging provides biologic information on the tumor and has proved useful for staging, surveillance, and radiation treatment planning. However, the utility of PET/CT imaging in management of gastric cancer is unclear (5). Gastric cancer has low FDG avidity compared to other gastrointestinal malignancies (6). It is not routinely recommended in the staging of newly-diagnosed gastric cancer, although it may provide some benefit for evaluation of regional lymph node involvement (6,7). However, PET/CT imaging may have some utility for gastric cancer in the setting of recurrent disease or response to chemotherapy (8,9).
In contrast to its role in gastric cancer, PET/CT imaging has shown to be an effective tool in the detection of metastatic liver disease, both for colorectal and non-colorectal malignancies (10). As a result, utilization of PET/CT imaging may improve target delineation for liver metastases compared to MRI (4). In addition, two planning studies have suggested that 4D PET/CT may provide added benefit for liver SBRT planning and target delineation compared to non-gated PET/CT imaging (4,11).
Our case emphasizes the advantages and disadvantages of PET/CT imaging in the target delineation and treatment planning of SBRT for metastatic gastric cancer in the liver. PET/CT imaging was able to identify areas of metastatic disease not visible on CT imaging. However, it was not able to delineate an area of disease (possibly due to mucin production from the metastatic gastric cancer cells) that was visible on CT imaging. Therefore, our case highlights the importance of utilizing all available imaging modalities when performing SBRT for liver metastases in order to maximize local disease control.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Gomez D, Lobo DN. Malignant liver tumours. Surgery 2011;29:632-9.
- Høyer M, Swaminath A, Bydder S, et al. Radiotherapy for liver metastases: a review of evidence. Int J Radiat Oncol Biol Phys 2012;82:1047-57. [Crossref] [PubMed]
- Wurm RE, Gum F, Erbel S, et al. Image guided respiratory gated hypofractionated Stereotactic Body Radiation Therapy (H-SBRT) for liver and lung tumors: Initial experience. Acta Oncol 2006;45:881-9. [Crossref] [PubMed]
- Bundschuh RA, Andratschke N, Dinges J, et al. Respiratory gated [18F]FDG PET/CT for target volume delineation in stereotactic radiation treatment of liver metastases. Strahlenther Onkol 2012;188:592-8. [Crossref] [PubMed]
- Hopkins S, Yang GY. FDG PET imaging in the staging and management of gastric cancer. J Gastrointest Oncol 2011;2:39-44. [PubMed]
- Lambrecht M, Haustermans K. Clinical evidence on PET-CT for radiation therapy planning in gastro-intestinal tumors. Radiother Oncol 2010;96:339-46. [Crossref] [PubMed]
- Dassen AE, Lips DJ, Hoekstra CJ, et al. FDG-PET has no definite role in preoperative imaging in gastric cancer. Eur J Surg Oncol 2009;35:449-55. [Crossref] [PubMed]
- Bilici A, Ustaalioglu BB, Seker M, et al. The role of 18F-FDG PET/CT in the assessment of suspected recurrent gastric cancer after initial surgical resection: can the results of FDG PET/CT influence patients' treatment decision making? Eur J Nucl Med Mol Imaging 2011;38:64-73. [Crossref] [PubMed]
- Shimada H, Okazumi S, Koyama M, et al. Japanese Gastric Cancer Association Task Force for Research Promotion: clinical utility of 18F-fluoro-2-deoxyglucose positron emission tomography in gastric cancer. A systematic review of the literature. Gastric Cancer 2011;14:13-21. [Crossref] [PubMed]
- Chua SC, Groves AM, Kayani I, et al. The impact of 18F-FDG PET/CT in patients with liver metastases. Eur J Nucl Med Mol Imaging 2007;34:1906-14. [Crossref] [PubMed]
- Riou O, Serrano B, Azria D, et al. Integrating respiratory-gated PET-based target volume delineation in liver SBRT planning, a pilot study. Radiat Oncol 2014;9:127. [Crossref] [PubMed]


