Extramedullary plasmacytoma of the gallbladder diagnosed by endoscopic ultrasound fine needle aspiration (EUS-FNA)
Introduction
Plasma cell myeloma (PCM) is a common hematologic malignancy that affects 24,000 patients in the US population each year and makes up 1.4% of all new cancer diagnoses. The median age at diagnosis is 69 years (1). It is characterized by intramedullary clonal plasma cells, with extramedullary disease traditionally a rare phenomenon that occurs in 4-6% of cases. However, extramedullary disease is increasingly common both at diagnosis and in follow-up as greater use of imaging modalities becomes standard of care. The presence of extramedullary disease carries prognostic significance as patients with extramedullary disease demonstrate decreased survival compared to those with intramedullary disease alone (2,3). The most common locations for extramedullary disease are the gastrointestinal tract, pleura, testis, skin, peritoneum, liver, endocrine glands and lymph nodes (3,4). The gallbladder is an extremely rare site of extramedullary disease. We report a case of extramedullary PCM of the gallbladder, with confirmed histopathologic diagnosis established by endoscopic ultrasound (EUS)-guided fine needle aspiration (FNA).
Case report
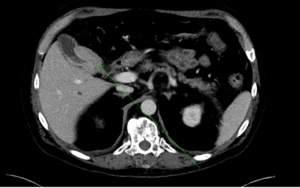
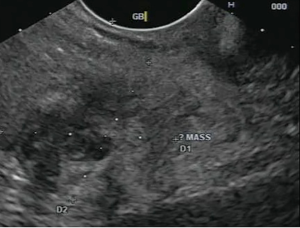
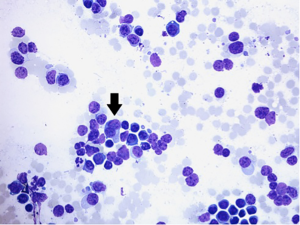
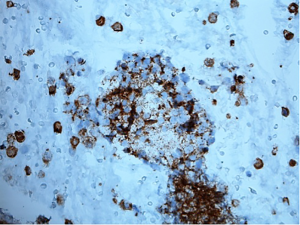
A 53-year-old male with multiple myeloma was admitted to Duke University Medical Center in December 2013 with worsening diarrhea, fatigue, nausea and anorexia. He had previously undergone autologous stem cell transplant a year and a half prior to presentation, with subsequent allogeneic stem cell transplant 14 months prior to presentation. His post stem-cell transplant course had been complicated by graft versus host disease affecting the skin, gastrointestinal tract, and liver. Three weeks prior to presentation, he underwent a skin biopsy that was interpreted as favoring graft versus host disease. Because of the skin biopsy results, he was placed on prednisone 100 mg twice daily, which was tapered to 80 mg twice daily one week prior to admission. Shortly thereafter he developed the above symptoms of diarrhea, fatigue, nausea and anorexia that prompted his admission. Admission labs were notable for a mild thrombocytopenia (110×109/L) and mild leukocytosis (13×109/L) with a mild hyponatremia (131 mmol/L) and abnormal liver chemistries of ALT 630 U/L, AST 195 U/L, alkaline phosphatase 252 U/L, bilirubin 2.1 mg/dL, and albumin 2.1 g/dL. He and was started on total parenteral nutrition and intravenous solumedrol for presumed graft versus host disease. Admission chest radiograph showed new hazy bilateral pulmonary opacities. The chest radiograph findings prompted a CT scan of his chest that incidentally demonstrated a new mass in the gallbladder (Figure 1). Surgical consultation recommended observation and treatment of his graft versus host disease with possible interval cholecystectomy due to his acute illness. Subsequently an abdominal MRI was obtained and again demonstrated a 1.9 cm thick circumferential enhancing mass involving the neck and body of the gallbladder. Eleven days following admission, he underwent an EUS, which showed an irregular heterogeneous mass that appeared to fill the entire gallbladder and measured 37 mm × 23 mm in maximal cross sectional diameter. There was sonographic evidence suggesting invasion into the gallbladder wall as manifested by intraluminal growth (Figure 2). FNA was performed using a transduodenal approach with a 25 gauge Wilson-Cook Echotip Procore ultrasound biopsy needle. The cytopathology specimen was diagnostic of a plasma cell neoplasm, showing morphologically atypical plasma cells (Figure 3). These cells were positive by immunohistochemistry for CD138 (Figure 4), MUM-1, and kappa light chain (weak), and negative for CD56, CD79a, CD20, CD3, lambda, and CD5.
The decision was made to start radiation therapy (2,400 cGy in 8 fractions) since the patient had developed cholangitis and was not a surgical candidate. Follow up imaging with MRI and EUS demonstrated a decrease in size of the mass following radiation.
Discussion
Extramedullary spread of PCM is traditionally a rare occurrence, but is becoming increasingly more common due to increased imaging and improved survival in myeloma patients (2). Nevertheless, involvement of the gallbladder is quite uncommon, and limited to sporadic case reports only. Heckmann et al. describe a 70-year-old man with PCM who underwent abdominal ultrasound and had multiple hypodense liver lesions and hyperdense polypoid masses of the gallbladder that developed into concentric wall thickening 5 weeks later on abdominal CT; histologic diagnosis was confirmed by liver biopsy, but tissue was not obtained from the gallbladder mass (5). Schuster et al. report a 66-year-old man with a history of solitary plasmacytoma who presented with acute cholecystitis and was found to have metastatic infiltration of the gallbladder by an extramedullary plasmacytoma (EMP) on cholecystectomy specimen (6). Hwang et al. describe a 63-year-old woman with CBD obstruction who underwent cholecystectomy with lymph node dissection and was found to have gallbladder plasmacytoma, but no evidence of underlying PCM on bone marrow biopsy; she received six cycles of CHOP and was disease free at 8 years (7). Likewise, Majerović et al. and Kondo et al. report cases of gallbladder plasmacytoma that were treated surgically and showed no evidence of recurrent disease or of underlying PCM (8,9). The association, or lack thereof, with underlying PCM is important, for while PCM complicated by EMP carries a poor prognosis, EMP that does not occur in conjunction with underlying myeloma is a biologically different disease with indolent clinical course (2).
In conclusion, we have described a case of EMP of the gallbladder associated with underlying PCM. This is the first case of which we are aware to have been diagnosed using EUS and the first to be confirmed by cytopathologic analysis. The disease can manifest as acute cholecystitis, biliary obstruction, or be asymptomatic as is the case in our patient. While treatment of EMP is traditionally surgical, we demonstrated successful disease control using localized radiation therapy. In patients with better functional status, surgical resection plus adjuvant chemotherapy or autologous stem cell transplant also seem to be reasonable options.
Acknowledgements
Disclosure: The authors declare no conflict of interest.
References
- Surveillance, Epidemiology, and End Results (SEER) Program Data (1973-2011). April 2014 ed: National Cancer Institute, DCCPS, Surveillance Research Program, Surveillance Systems Branch, Released April 2014, based on the November 2013 submission.
- Wirk B, Wingard JR, Moreb JS. Extramedullary disease in plasma cell myeloma: the iceberg phenomenon. Bone Marrow Transplant 2013;48:10-8. [PubMed]
- Varettoni M, Corso A, Pica G, et al. Incidence, presenting features and outcome of extramedullary disease in multiple myeloma: a longitudinal study on 1003 consecutive patients. Ann Oncol 2010;21:325-30. [PubMed]
- Damaj G, Mohty M, Vey N, et al. Features of extramedullary and extraosseous multiple myeloma: a report of 19 patients from a single center. Eur J Haematol 2004;73:402-6. [PubMed]
- Heckmann M, Uder M, Grgic A, et al. Extraosseous manifestation of multiple myeloma with unusual appearance in computed tomography--case report. Rontgenpraxis 2008;56:249-53. [PubMed]
- Schuster D, Klosterhalfen B, Fiedler C, et al. Metastasis of medullary plasmocytoma as the cause of acute cholecystitis. Dtsch Med Wochenschr 2007;132:612-5. [PubMed]
- Hwang DW, Lim CS, Jang JY, et al. Primary hematolymphoid malignancies involving the extrahepatic bile duct or gallbladder. Leuk Lymphoma 2010;51:1278-87. [PubMed]
- Kondo H, Kainuma O, Itami J, et al. Extramedullary plasmacytoma of maxillary sinus with later involvement of the gall bladder and subcutaneous tissues. Clin Oncol (R Coll Radiol) 1995;7:330-1. [PubMed]
- Majerović M, Bogdanić B, Drinković N, et al. Extramedullary plasmacytoma imitating neoplasm of the gallbladder fossa after cholecystectomy. Coll Antropol 2012;36:331-3. [PubMed]