
Surgical management of pancreatic neuroendocrine liver metastases
Introduction
Pancreatic neuroendocrine tumors (PNET) are a heterogeneous group of neoplasms that arise from pancreatic islet cells (1). Even among well-differentiated tumors, PNET can have variable clinical presentation, behavior and prognosis. Among patients with PNETs, the liver is the most common site of metastasis, with approximately 28–77% of patients developing neuroendocrine liver metastases (NELM) in their lifetime (2,3). The development of pancreatic NELM (P-NELM) can be associated with significant detriments in quality and quantity of life. Patients with liver metastases can experience debilitating carcinoid syndrome, as well as local complications such as biliary obstruction or liver insufficiency. In addition, the presence of P-NELM is one of the most significant negative prognostic factors among patients with PNETs. For these reasons, a clear understanding of the available therapeutic strategies for patients with P-NELM is critical to ensuring optimal outcomes. In this article, we review the surgical management of patients with P-NELM.
Hepatic resection
Outcomes
Although no randomized controlled trials have been performed (4), surgical resection of NELM is believed to afford the best long-term outcomes when feasible. Five-year OS can be as high as 60–80% among well selected patients undergoing resection (5). A large single-institution analysis of patients with P-NELM undergoing hepatic resection reported a median overall survival (OS) of 124 months and 5-, 10-, and 20-year OS rates of 67%, 51%, and 36%, respectively (6). These outcomes are more favorable compared with outcomes of patients treated with nonoperative approaches. For example, a retrospective multi-institutional review of 339 patients with NELM undergoing liver resection versus 414 patients receiving intra-arterial therapies noted an improved 5-year OS among patients who underwent surgery (74% vs. 30%, P<0.001) (7).
Several meta-analyses have attempted to confirm the benefit of surgery for NELM. For example, a 2012 systematic review reported pooled median 3-, 5- and 10-year OS of 83% (range, 63–100%), 70.5% (range, 31–100%), and 42% (range, 0–100%), respectively (8). A different systematic review noted significantly improved survival outcomes following liver resection compared with liver-directed therapies, chemotherapy, or no treatment (9). A meta-analysis of seven studies that included only patients with P-NELM noted improved symptom control and longer survival durations with hepatic resection versus non-surgical treatments (10). Nevertheless, the data were not uniformly concordant. A more recent systematic review “identified no robust evidence that a liver resection was superior to any other liver-directed therapies” to improve OS, progression-free survival (PFS), or quality of life (QOL) for patients with NELM (11,12).
In light of the excellent long-term outcomes observed in retrospective series of patients undergoing resection for NELM, and the fact that a prospective randomized controlled trial is unlikely to be conducted, surgery for appropriately selected patients in the context of a multidisciplinary neuroendocrine program is recommended. A recent international Working Group on Neuroendocrine Liver Metastases similarly concluded that the use of liver resection is the preferred therapeutic strategy for patients with NELM when feasible (13). Similar guideline recommendations have been made by the North American Neuroendocrine Tumor Society (NANETS) (14) and the European Neuroendocrine Tumor Society (ENETS) (2) as well as others (15-17).
Prognostic factors
A better understanding of prognostic factors associated with outcomes following surgery may assist with patient selection for liver resection. To this point, multiple studies have evaluated factors associated with long-term outcomes. For example, Glazer et al. reported that synchronous disease, nonfunctional hormonal status, and extra-hepatic disease were adversely associated with long-term outcomes, while Spolverato et al. reported that extrahepatic disease and tumor grade were associated with worse prognosis (12,18). In a systematic review, Saxena et al. noted that poor histologic grade, extrahepatic disease, and a macroscopically incomplete resection were associated with worse long-term outcomes (8). While extrahepatic metastatic disease is one of the most consistent negative prognostic factors observed across studies, hepatic resection in the setting of low-volume indolent bone metastases may be appropriate (19). In addition, synchronous disease is associated with higher recurrence rates compared with metachronous disease (20).
Interestingly, most, but not all studies, have found that R1 margin status is not associated with worse outcomes (18,21-23). The lack of prognostic impact of R1 margin status may be due to the relative expansive or well-encapsulated behavior of neuroendocrine metastases versus the more infiltrative nature of other liver malignancies. For this reason, parenchymal sparing approaches including hepatic enucleations may be considered, as such technical approaches appear to have similar long-term outcomes (24).
Debulking
Many surgeons have argued for expanding the role of surgery for NELM to patients in which complete macroscopic removal of all disease is not possible (25). Indeed, several series have documented good long-term outcomes and symptom control for cytoreductive surgery (i.e., debulking) in which 80–90% of tumors are removed. Ejaz et al. reported a 5-year OS of 60% among 179 patients with NELM who underwent non-curative cytoreductive surgery (26). Similarly, Sarmiento et al. reported a 5-year OS of 61%, as well as excellent symptom control among 170 patients undergoing hepatic resection with 90% debulking (27). Recently, Scott et al. reported on the outcomes of patients with NELM undergoing debulking and reported that patients who had >70% of metastatic disease resected had improved OS (median 134 vs. 38 months) (28). Similarly, Morgan et al. noted comparable survival outcomes among patients undergoing 70%, 90%, and 100% of tumor volume and therefore also proposed changing the threshold for cytoreductive surgery to 70% (25).
The role of hepatic resection for P-NELM is similarly unclear in the setting of an unresectable primary tumor. Given that most patients with NELM eventually die from complications of their liver disease and not local complications of the primary, aggressive management of the NELM may be indicated even in the setting of an unresectable primary PNET. In fact, a recent multi-institutional, propensity-matched, retrospective analysis noted similar long-term OS among patients with NELM who did and did not undergo primary tumor resection (PTR) (29).
Concomitant ablation
Ablative procedures may be used alone or in combination with surgical resection to address liver metastases. Concomitant ablation is mainly used in the setting of multifocal disease to assist with debulking of unresectable disease or to avoid extended hepatic resection. Taner et al. reported that ablation used in combination with resection resulted in 5- and 10-year OS of 80% and 59%, respectively (30). In a large multi-institutional analysis, ablative procedures were used in approximately 20% of patients undergoing surgery for NELM, which was not associated with an increased risk of recurrence or worse survival (31). Approximately 30% of resections for NELM performed in the United States include concomitant ablative procedures suggesting ablation is a routine part of surgical practice at most centers.
Several technical issues should be considered when utilizing concomitant ablative procedures. While radiofrequency ablation (RFA) has historically been the most commonly utilized technique, increasingly microwave ablation (MWA) is more often used and may be the preferred modality (32). Ablation should be used for small-intermediate sized tumors to preserve liver parenchyma or avoid the morbidity of major resection. Some caution should be given in combining ablation with extended hepatectomies as ablation in the future liver remnant could increase the risk for postoperative hepatic insufficiency (33). Care should also be exercised when ablating lesions close to hepatic veins (heat sink) or portal pedicles (biliary injury, liver ischemia).
Resection of the primary
For patients with synchronous resectable NELM, resection of the primary either in a staged or combined fashion is recommended. The decision for a staged versus combined approach is typically influenced by the location and number of the liver metastases as well as the primary tumor. Similar to the strategy employed for patients with synchronous colorectal liver metastases, minor liver resections can be combined with pancreatectomy; however, major liver resections should be more cautiously considered as pancreatoduodenectomy or complicated left-sided pancreatectomy may increase overall morbidity and mortality (34,35).
The role of PTR in the setting of unresectable metastatic disease is controversial. For midgut NETs, the goals of PTR include histological confirmation, resolution or prevention of obstruction, and potentially improvement of survival. A systematic review and meta-analysis of PTR in the setting of unresectable NELM suggested improvement in OS even in the setting of asymptomatic primary tumors (36) and this practice is generally recommended by both NANETS and ENETS for well-differentiated small intestine NETs (37,38). The decision to proceed with pancreatectomy in the setting of unresectable NELM can be more challenging, however, given the additional morbidity associated with pancreatic resection and the lower odds of symptom development from a pancreatic primary compared to an intestinal primary. Nevertheless, similar meta-analyses have suggested long-term survival gains associated with primary pancreatic tumor resection in the setting of unresectable NELM (39). However, no randomized controlled trials have been performed on this topic and thus this evidence needs to be interpreted with caution. While significant methodologic biases likely confound the interpretation of all retrospective studies on this topic, the data supports at least consideration of PTR especially for healthy patients with low tumor burden.
Recurrent disease
Despite an excellent OS following surgical resection of NELM, most patients will experience recurrence of their disease, most commonly in the liver. In fact, the rate of disease recurrence after hepatic resection approaches 50–95% (8,31) although statistical cure is possible among patients who undergo liver-directed surgery (40). Among patients who develop intrahepatic recurrence, >70% will occur within 3 years of surgery, and approximately 40% within one year (23,41). In other primary and metastatic tumors of the liver, multiple studies have highlighted the safety and relative efficacy of repeat hepatectomy for recurrent disease (42,43). Repeat hepatic resection for recurrent NELM is feasible and associated with good long-term survival outcomes among well-selected patients (44). Non-surgical therapies are alternative options for recurrent liver metastases.
Surgical considerations
Patient selection
Careful patient selection is imperative for optimizing short- and long-term outcomes of hepatic resection (Box 1). Patients should be evaluated along three domains: anatomic, biologic, and physiologic. First, in general, patient’s liver metastases should be amenable to complete or >90% resection. As discussed above, cytoreductive surgery when >70% of disease can be debulked may be considered especially for patients with functional tumors or with carcinoid syndrome. Hepatic resection should leave a future liver remnant with at least two contiguous segments characterized by intact venous, arterial, portal venous, and biliary drainage that comprises at least 20% of standardized liver volume. Minimal to no extrahepatic disease should be present and, in general, the primary tumor should be resectable. Finally, patients should be healthy enough to undergo safely major hepatic resection with acceptable morbidity. For those patients with prohibitive or borderline comorbidities that cannot be optimized, other liver-directed therapies should be prioritized.

Full table
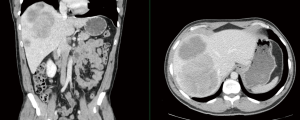
General guidelines exist for selecting NELM patients for hepatic resection. ENETS recommends the following criteria: R0 resection feasible, grade 1 or 2 tumors, acceptable perioperative morbidity (~30%) and mortality (<5%), absence of right heart failure, absence of extra-hepatic disease, and no peritoneal carcinomatosis (37). However, given the unique biology of NETs, it is not uncommon for indications for surgery to fall outside these criteria. A common “off-label” indication for resection is palliation of large tumors in the setting of unresectable but low volume liver metastases (Figure 1). Another common “off-label” indication would be complete resection of NELM in the setting of minimal extrahepatic disease which would otherwise be treated with observation or somatostatin analogs (SSAs) alone without the liver burden. In both of these scenarios, surgery is an integral component of, and actually facilitates, the multidisciplinary management of metastatic NETs.
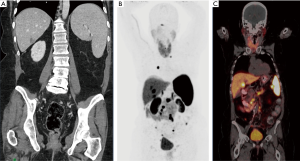
Prior to liver resection, patients should undergo a thorough medical history and physical examination with particular attention to symptoms of carcinoid syndrome as well as signs of liver insufficiency. Thin-slice triple-phase computed tomography (CT) and/or magnetic resonance imaging (MRI) is critical for surgical planning. In addition, all patients should have preoperative somatostatin receptor positron emission tomography (PET), most commonly 68Ga-DOTATATE, to assess for extrahepatic disease and confirm presence of somatostatin receptors (45). Previous studies have demonstrated that neuroendocrine PET commonly identifies additional hepatic and/or extrahepatic metastases that may influence treatment decision making (46,47) (Figure 2). Laboratory tests should include basic complete blood count, complete metabolic profile, and coagulation profile as well as neuroendocrine hormones such as chromogranin A as indicated. Patient health and functional status should be assessed and optimized as needed.
Perioperative management
With advances in patient selection, anesthetic medicine, and perioperative care, the safety of complex liver surgery is increasing (48). However, several specific considerations should be undertaken for patients with neuroendocrine tumors. First, patients with NELM are at risk for carcinoid crisis (49). While the incidence of carcinoid crisis is low, some clinicians recommend the use of perioperative octreotide given that the disadvantages and cost of administration are relatively low and the consequences of developing a carcinoid crisis potentially catastrophic. Controversy exists over the appropriate perioperative dosage as well as its actual efficacy (50). In addition, some surgeons do not routinely administer octreotide for patients with asymptomatic PNET NELM, especially those patients with a low burden of disease.
Standard surgical principles of liver surgery apply for NELM, namely prioritizing low central venous pressure anesthesia, minimizing blood loss, knowledge of liver anatomy, generous use of intraoperative ultrasound, as well as safe parenchymal transection techniques and practices to avoid bile leakage. For combined operations, liver-directed surgery is often prioritized to be completed first in order to take advantage of low central venous pressure. Minimally invasive approaches are increasingly being used for hepatic surgery and are acceptable for certain patients with NELM (51). Enhanced recovery programs are also an important part of complex hepatic surgery programs that hasten recovery and shorten length of hospital stay; these should be implemented for patients with NELM (52).
Given the frequent multifocality of NELM and the fact that preoperative imaging often underestimates the number of tumors, the liver should be thoroughly evaluated for occult metastases. Intraoperative ultrasound is essential to order identify all visible tumors; it is also useful for image-guided ablation. Novel methods of intraoperative localization are currently being evaluated (53). Following surgery, patients should be followed with cross-sectional imaging and tumor markers every 3–12 months (17).
Neoadjuvant and adjuvant treatment strategies
Given the high rate of recurrence following hepatic resection of NELM (8,31), the use of multimodality therapeutic strategies to improve patient selection for surgery and reduce the development of recurrent disease are appealing. Although significant progress has been in made in recent years with regards to medical therapies for patients with unresectable or progressive metastatic disease (54-57), little research has been conducted on strategies for preventing recurrence in high risk NETs. This is an important goal, though, since tumor recurrence is one key factor independently associated with disease-specific mortality among patients with non-functional well-differentiated NETs, and perhaps the only one that is potentially modifiable (58). Indeed, a recent international consensus conference on NELM highlighted the need for clinical trials on adjuvant therapy strategies (13).
While few studies have investigated the use of adjuvant therapy following resection of NELM (59), a few studies have reported on the use of neoadjuvant therapies for patients with P-NELM. Most case reports have highlighted the use of non-surgical therapies to downstage patients in order to facilitate resection of NELM (60). Cloyd et al. reported a single institution retrospective experience of neoadjuvant fluorouracil, doxorubicin, and streptozocin prior to the resection of P-NELM and reported improved OS compared with immediate surgery, especially among patients with synchronous disease (61). Preliminary evidence suggests that the orally-available regimen capecitabine and temozolomide is also associated with excellent response rates and long-term outcomes prior to resection of P-NELM (62). Additional research is needed to clarify the potential benefits of neoadjuvant and adjuvant therapies for patients with P-NELM, as well as appropriate selection criteria.
Liver-directed therapies
For patients who are not surgical candidates, due to either patient performance status or burden of liver disease, several liver-directed therapies are available to assist with locoregional control, extend PFS, and improve symptoms of carcinoid syndrome. For those patients with oligometastatic disease, percutaneous ablation, either radiofrequency or microwave, can be used. Outcomes are optimized among patients with tumors <3 cm away from major vascular structures. For patients with multiple tumors, especially bilobar disease, transarterial therapies are usually indicated (63). Transarterial embolization (TAE) can be performed bland (64) or in conjunction with chemotherapy (65) (i.e., transarterial chemoembolization, TACE) or yttrium-90 (66) (transarterial radioembolization, TARE). There is currently insufficient evidence to conclude superiority of one transarterial approach over others, though some evidence suggests conventional TACE may result in improved long-term outcomes compared with TARE, potentially at the expense of increased early complications (67).
Despite the efficacy of liver-directed therapies in controlling symptoms and prolonging survival, in general, this therapeutic approach is not curative. Therefore, liver transplantation (LT) has long held appeal as a potentially curative surgical option for patients with unresectable NELM (68). However, given the shortage of available organs, the frequency of extrahepatic metastatic disease, and incomplete evidence to define its role in the management of metastatic NETs, LT has been utilized relatively infrequently for NELM. Recent systematic reviews have suggested a 5-year OS of >50%, but the data are based on retrospective studies with significant selection biases and inter-study heterogeneity (2,69). General guidelines for the selection of patients with NELM for LT have been previously outlined (70). However, additional larger scale research is needed to define the optimal indications for and outcomes of LT for P-NELM before it can be used more routinely.
Systemic therapies
Multiple systemic therapy options exist for patients with metastatic PNET (71). While often prioritized for those patients with advanced or progressive disease, systemic therapies can also be utilized for individuals with isolated NELM in addition to or in lieu of liver-directed therapies. Often the initial treatment for patients with low-volume well-differentiated tumors, SSAs lead to improved PFS and effectively control the symptoms of hormonal overproduction (54,72). Although cytotoxic chemotherapy is more commonly used for patients with high grade neuroendocrine carcinoma, it has been recognized for several decades that well-differentiated PNETs respond to streptozocin-based chemotherapy regimens (73). More recent evidence suggests that the combination capecitabine/temozolomide is efficacious and has the advantage of being orally available (74). Furthermore, advances in the molecular understanding of PNETs have led to the development of multiple novel targeted therapies (71). For example, both sunitinib malate, an oral, small molecule, multitargeted tyrosine kinase inhibitor initially approved for the treatment of advanced renal cell carcinoma and refractory gastrointestinal stromal tumors, as well as everolimus, an oral inhibitor of the mammalian target of rapamycin (mTOR), are specifically approved for use in metastatic PNETs (55,56). Finally, peptide receptor radionuclide therapy (PRRT) delivers SSA-bound radionuclides selectively to somatostatin receptor positive tumors and is now approved for use in the United States based on the results of the NETTER-1 trial (57). Ongoing research is currently investigating novel radiopharmaceuticals as well as targeted therapies, checkpoint inhibitors, and oncocytic viruses (71).
Conclusions
In conclusion, PNETs are a heterogeneous neoplasm occurring with rising frequency that commonly metastasize to the liver. Patients with P-NELM require a comprehensive evaluation and a multidisciplinary approach. For eligible patients, hepatic resection is recommended as it is associated with the best long-term outcomes and excellent QOL. Surgical resection may be appropriate even in the setting of an unresectable primary tumor, low-volume indolent extrahepatic disease, or when only >70% of liver metastases can be debulked. However, prospective clinical trials are scarce and significant evidence gaps exist. Fortunately, multiple liver-directed and systemic treatment options exist for those patients who are not surgical candidates or develop recurrent disease. A systematic approach in a multi-disciplinary setting is likely to afford the best long-term outcomes for patients with P-NELM. Ongoing research is needed to determine the optimal patient selection for hepatic surgery as well as the ideal treatment sequencing for those patients with P-NELM.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Callisia N. Clarke, Douglas B. Evans) for the series “Pancreatic Neuroendocrine Tumors” published in Journal of Gastrointestinal Oncology. The article was sent for external peer review organized by the Guest Editors and the editorial office.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jgo.2019.11.02). The series “Pancreatic Neuroendocrine Tumors” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Cloyd JM, Poultsides GA. Non-functional neuroendocrine tumors of the pancreas: Advances in diagnosis and management. World J Gastroenterol 2015;21:9512-25. [Crossref] [PubMed]
- Pavel M, Baudin E, Couvelard A, et al. ENETS Consensus Guidelines for the Management of Patients with Liver and Other Distant Metastases from Neuroendocrine Neoplasms of Foregut, Midgut, Hindgut, and Unknown Primary. Neuroendocrinology 2012;95:157-76. [Crossref] [PubMed]
- Yao JC, Hassan M, Phan A, et al. One Hundred Years After “Carcinoid”: Epidemiology of and Prognostic Factors for Neuroendocrine Tumors in 35,825 Cases in the United States. J Clin Oncol 2008;26:3063-72. [Crossref] [PubMed]
- Gurusamy KS, Ramamoorthy R, Sharma D, et al. Liver resection versus other treatments for neuroendocrine tumours in patients with resectable liver metastases. Cochrane Hepato-Biliary Group, editor. Cochrane Database Syst Rev [Internet]. 2009 Apr 15 [cited 2019 Oct 21]; Available online: http://doi.wiley.com/10.1002/14651858.CD007060.pub2
- Fairweather M, Swanson R, Wang J, et al. Management of Neuroendocrine Tumor Liver Metastases: Long-Term Outcomes and Prognostic Factors from a Large Prospective Database. Ann Surg Oncol 2017;24:2319-25. [Crossref] [PubMed]
- Woltering EA, Voros BA, Beyer DT, et al. Aggressive Surgical Approach to the Management of Neuroendocrine Tumors: A Report of 1,000 Surgical Cytoreductions by a Single Institution. J Am Coll Surg 2017;224:434-47. [Crossref] [PubMed]
- Mayo SC, Pawlik TM. Surgical management of neuroendocrine tumors: treatment of localized and metastatic disease. Oncology (Williston Park) 2011;25:806-808, 810. [PubMed]
- Saxena A, Chua TC, Perera M, et al. Surgical resection of hepatic metastases from neuroendocrine neoplasms: A systematic review. Surg Oncol 2012;21:e131-41. [Crossref] [PubMed]
- Kaçmaz E, Heidsma CM, Besselink MGH, et al. Treatment of Liver Metastases from Midgut Neuroendocrine Tumours: A Systematic Review and Meta-Analysis. J Clin Med 2019. [Crossref] [PubMed]
- Yuan CH, Wang J, Xiu DR, et al. Meta-analysis of Liver Resection Versus Nonsurgical Treatments for Pancreatic Neuroendocrine Tumors with Liver Metastases. Ann Surg Oncol 2016;23:244-9. [Crossref] [PubMed]
- Lesurtel M, Nagorney DM, Mazzaferro V, et al. When should a liver resection be performed in patients with liver metastases from neuroendocrine tumours? A systematic review with practice recommendations. HPB (Oxford) 2015;17:17-22. [Crossref] [PubMed]
- Spolverato G, Vitale A, Ejaz A, et al. Net health benefit of hepatic resection versus intraarterial therapies for neuroendocrine liver metastases: A Markov decision model. Surgery 2015;158:339-48. [Crossref] [PubMed]
- Frilling A, Modlin IM, Kidd M, et al. Recommendations for management of patients with neuroendocrine liver metastases. Lancet Oncol 2014;15:e8-21. [Crossref] [PubMed]
- Kunz PL, Reidy-Lagunes D, Anthony LB, et al. Consensus Guidelines for the Management and Treatment of Neuroendocrine Tumors. Pancreas 2013;42:557-77. [Crossref] [PubMed]
- Singh S, Dey C, Kennecke H, et al. Consensus Recommendations for the Diagnosis and Management of Pancreatic Neuroendocrine Tumors: Guidelines from a Canadian National Expert Group. Ann Surg Oncol 2015;22:2685-99. [Crossref] [PubMed]
- Jin K, Xu J, Chen J, et al. Surgical management for non-functional pancreatic neuroendocrine neoplasms with synchronous liver metastasis: A consensus from the Chinese Study Group for Neuroendocrine Tumors (CSNET). Int J Oncol 2016;49:1991-2000. [Crossref] [PubMed]
- NCCN Clinical Practice Guidelines in Oncology. Neuroendocrine and Adrenal Tumors. 2019 Mar:1-135. Available online: https://www.nccn.org/professionals/physician_gls/pdf/neuroendocrine.pdf
- Glazer ES, Tseng JF, Al-Refaie W, et al. Long-term survival after surgical management of neuroendocrine hepatic metastases. HPB (Oxford) 2010;12:427-33. [Crossref] [PubMed]
- Croome KP, Burns JM, G, Que F, et al. Hepatic Resection for Metastatic Neuroendocrine Cancer in Patients with Bone Metastases. Ann Surg Oncol 2016;23:3693-8. [Crossref] [PubMed]
- Zhang XF, Beal EW, Weiss M, et al. Timing of disease occurrence and hepatic resection on long-term outcome of patients with neuroendocrine liver metastasis. J Surg Oncol 2018;117:171-81. [Crossref] [PubMed]
- Elias D, Lasser P, Ducreux M, et al. Liver resection (and associated extrahepatic resections) for metastatic well-differentiated endocrine tumors: A 15-year single center prospective study. Surgery 2003;133:375-82. [Crossref] [PubMed]
- Farley HA, Pommier RF. Surgical Treatment of Small Bowel Neuroendocrine Tumors. Hematol Oncol Clin North Am 2016;30:49-61. [Crossref] [PubMed]
- Zhang XF, Beal EW, Chakedis J, et al. Early Recurrence of Neuroendocrine Liver Metastasis After Curative Hepatectomy: Risk Factors, Prognosis, and Treatment. J Gastrointest Surg 2017;21:1821-30. [Crossref] [PubMed]
- Maxwell JE, Sherman SK, O'Dorisio TM, et al. Liver-directed surgery of neuroendocrine metastases: What is the optimal strategy? Surgery 2016;159:320-33. [Crossref] [PubMed]
- Morgan RE, Pommier SJ, Pommier RF. Expanded criteria for debulking of liver metastasis also apply to pancreatic neuroendocrine tumors. Surgery 2018;163:218-25. [Crossref] [PubMed]
- Ejaz A, Reames BN, Maithel S, et al. Cytoreductive debulking surgery among patients with neuroendocrine liver metastasis: a multi-institutional analysis. HPB (Oxford) 2018;20:277-84. [Crossref] [PubMed]
- Sarmiento JM, Heywood G, Rubin J, et al. Surgical treatment of neuroendocrine metastases to the liver: a plea for resection to increase survival. J Am Coll Surg 2003;197:29-37. [Crossref] [PubMed]
- Scott AT, Breheny PJ, Keck KJ, et al. Effective cytoreduction can be achieved in patients with numerous neuroendocrine tumor liver metastases (NETLMs). Surgery 2019;165:166-75. [Crossref] [PubMed]
- Xiang JX, Zhang XF, Beal EW, et al. Hepatic Resection for Non-functional Neuroendocrine Liver Metastasis: Does the Presence of Unresected Primary Tumor or Extrahepatic Metastatic Disease Matter? Ann Surg Oncol 2018;25:3928-35. [Crossref] [PubMed]
- Taner T, Atwell TD, Zhang L, et al. Adjunctive radiofrequency ablation of metastatic neuroendocrine cancer to the liver complements surgical resection. HPB (Oxford) 2013;15:190-5. [Crossref] [PubMed]
- Mayo SC, de Jong MC, Pulitano C, et al. Surgical Management of Hepatic Neuroendocrine Tumor Metastasis: Results from an International Multi-Institutional Analysis. Ann Surg Oncol 2010;17:3129-36. [Crossref] [PubMed]
- Vogl TJ, Naguib NN, Zangos S, et al. Liver metastases of neuroendocrine carcinomas: Interventional treatment via transarterial embolization, chemoembolization and thermal ablation. Eur J Radiol 2009;72:517-28. [Crossref] [PubMed]
- Mizuno T, Cloyd JM, Omichi K, et al. Two-Stage Hepatectomy vs One-Stage Major Hepatectomy with Contralateral Resection or Ablation for Advanced Bilobar Colorectal Liver Metastases. J Am Coll Surg 2018;226:825-34. [Crossref] [PubMed]
- Serrano Aybar PE, Bogach J, Wang J, et al. Simultaneous versus staged resection for synchronous colorectal cancer liver metastases: A population-based cohort study. J Clin Oncol 2019;37:abstr 3612.
- Serrano PE, Gafni A, Parpia S, et al. Simultaneous resection of colorectal cancer with synchronous liver metastases (RESECT), a pilot study. Int J Surg Protoc 2018;8:1-6. [Crossref] [PubMed]
- Tsilimigras DI, Ntanasis-Stathopoulos I, Kostakis ID, et al. Is Resection of Primary Midgut Neuroendocrine Tumors in Patients with Unresectable Metastatic Liver Disease Justified? A Systematic Review and Meta-Analysis. J Gastrointest Surg 2019;23:1044-54. [Crossref] [PubMed]
- O’Toole D, Kianmanesh R, Caplin M. ENETS 2016 Consensus Guidelines for the Management of Patients with Digestive Neuroendocrine Tumors: An Update. Neuroendocrinology 2016;103:117-8. [Crossref] [PubMed]
- Howe JR, Cardona K, Fraker DL, et al. The Surgical Management of Small Bowel Neuroendocrine Tumors: Consensus Guidelines of the North American Neuroendocrine Tumor Society. Pancreas 2017;46:715-31. [Crossref] [PubMed]
- Zhou B, Zhan C, Ding Y, et al. Role of palliative resection of the primary pancreatic neuroendocrine tumor in patients with unresectable metastatic liver disease: a systematic review and meta-analysis. Onco Targets Ther 2018;11:975-82. [Crossref] [PubMed]
- Bagante F, Spolverato G, Merath K, et al. Neuroendocrine liver metastasis: The chance to be cured after liver surgery. J Surg Oncol 2017;115:687-95. [Crossref] [PubMed]
- Xiang JX, Zhang XF, Weiss M, et al. Early recurrence of well-differentiated (G1) neuroendocrine liver metastasis after curative-intent surgery: Risk factors and outcome: XIANG ET AL. J Surg Oncol 2018;118:1096-104. [Crossref] [PubMed]
- Minagawa M, Makuuchi M, Takayama T, et al. Selection Criteria for Repeat Hepatectomy in Patients With Recurrent Hepatocellular Carcinoma. Ann Surg 2003;238:703-10. [Crossref] [PubMed]
- Adam R, Bismuth H, Castaing D, et al. Repeat hepatectomy for colorectal liver metastases. Ann Surg 1997;225:51-60; discussion 60-2. [Crossref] [PubMed]
- Spolverato G, Bagante F, Aldrighetti L, et al. Management and outcomes of patients with recurrent neuroendocrine liver metastasis after curative surgery: An international multi-institutional analysis. J Surg Oncol 2017;116:298-306. [Crossref] [PubMed]
- Hope TA, Bergsland EK, Bozkurt MF, et al. Appropriate Use Criteria for Somatostatin Receptor PET Imaging in Neuroendocrine Tumors. J Nucl Med 2018;59:66-74. [Crossref] [PubMed]
- Koopmans KP, Neels OC, Kema IP, et al. Improved staging of patients with carcinoid and islet cell tumors with 18F-dihydroxy-phenyl-alanine and 11C-5-hydroxy-tryptophan positron emission tomography. J Clin Oncol 2008;26:1489-95. [Crossref] [PubMed]
- Hofman MS, Kong G, Neels OC, et al. High management impact of Ga-68 DOTATATE (GaTate) PET/CT for imaging neuroendocrine and other somatostatin expressing tumours. J Med Imaging Radiat Oncol 2012;56:40-7. [Crossref] [PubMed]
- Cloyd JM, Mizuno T, Kawaguchi Y, et al. Comprehensive Complication Index Validates Improved Outcomes Over Time Despite Increased Complexity in 3707 Consecutive Hepatectomies. Ann Surg 2020;271:724-31. [Crossref] [PubMed]
- Kimbrough CW, Beal EW, Dillhoff ME, et al. Influence of carcinoid syndrome on the clinical characteristics and outcomes of patients with gastroenteropancreatic neuroendocrine tumors undergoing operative resection. Surgery 2019;165:657-63. [Crossref] [PubMed]
- Condron ME, Pommier SJ, Pommier RF. Continuous infusion of octreotide combined with perioperative octreotide bolus does not prevent intraoperative carcinoid crisis. Surgery 2016;159:358-65. [Crossref] [PubMed]
- Kandil E, Noureldine SI, Koffron A, et al. Outcomes of laparoscopic and open resection for neuroendocrine liver metastases. Surgery 2012;152:1225-31. [Crossref] [PubMed]
- Melloul E, Hübner M, Scott M, et al. Guidelines for Perioperative Care for Liver Surgery: Enhanced Recovery After Surgery (ERAS) Society Recommendations. World J Surg 2016;40:2425-40. [Crossref] [PubMed]
- El Lakis M, Gianakou A, Nockel P, et al. Radioguided Surgery With Gallium 68 Dotatate for Patients With Neuroendocrine Tumors. JAMA Surg 2019;154:40-5. [Crossref] [PubMed]
- Caplin ME, Pavel M, Ćwikła JB, et al. Lanreotide in Metastatic Enteropancreatic Neuroendocrine Tumors. N Engl J Med 2014;371:224-33. [Crossref] [PubMed]
- Raymond E, Dahan L, Raoul JL, et al. Sunitinib malate for the treatment of pancreatic neuroendocrine tumors. N Engl J Med 2011;364:501-13. [Crossref] [PubMed]
- Yao JC, Shah MH, Ito T, et al. Everolimus for advanced pancreatic neuroendocrine tumors. N Engl J Med 2011;364:514-23. [Crossref] [PubMed]
- Strosberg J, El-Haddad G, Wolin E, et al. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors. N Engl J Med 2017;376:125-35. [Crossref] [PubMed]
- Genç CG, Jilesen AP, Partelli S, et al. A New Scoring System to Predict Recurrent Disease in Grade 1 and 2 Nonfunctional Pancreatic Neuroendocrine Tumors. Ann Surg 2018;267:1148-54. [Crossref] [PubMed]
- Chung MH, Pisegna J, Spirt M, et al. Hepatic cytoreduction followed by a novel long-acting somatostatin analog: a paradigm for intractable neuroendocrine tumors metastatic to the liver. Surgery 2001;130:954-62. [Crossref] [PubMed]
- Perysinakis I, Aggeli C, Kaltsas G, et al. Neoadjuvant therapy for advanced pancreatic neuroendocrine tumors: an emerging treatment modality? Hormones (Athens) 2016;15:15-22. [Crossref] [PubMed]
- Cloyd JM, Omichi K, Mizuno T, et al. Preoperative Fluorouracil, Doxorubicin, and Streptozocin for the Treatment of Pancreatic Neuroendocrine Liver Metastases. Ann Surg Oncol 2018;25:1709-15. [Crossref] [PubMed]
- Squires M, Worth P, Konda B, et al. Neoadjuvant Capecitabine/Temozolomide for Locally Advanced or Metastatic Pancreatic Neuroendocrine Tumors. NANETS Annual Meeting; Boston, MA; 2019.
- Kennedy A, Bester L, Salem R, et al. Role of hepatic intra-arterial therapies in metastatic neuroendocrine tumours (NET): guidelines from the NET-Liver-Metastases Consensus Conference. HPB (Oxford) 2015;17:29-37. [Crossref] [PubMed]
- Elf AK, Andersson M, Henrikson O, et al. Radioembolization Versus Bland Embolization for Hepatic Metastases from Small Intestinal Neuroendocrine Tumors: Short-Term Results of a Randomized Clinical Trial. World J Surg 2018;42:506-13. [Crossref] [PubMed]
- Bloomston M, Al-Saif O, Klemanski D, et al. Hepatic artery chemoembolization in 122 patients with metastatic carcinoid tumor: lessons learned. J Gastrointest Surg 2007;11:264-71. [Crossref] [PubMed]
- Kennedy AS, Dezarn WA, McNeillie P, et al. Radioembolization for unresectable neuroendocrine hepatic metastases using resin 90Y-microspheres: early results in 148 patients. Am J Clin Oncol 2008;31:271-9. [Crossref] [PubMed]
- Do Minh D, Chapiro J, Gorodetski B, et al. Intra-arterial therapy of neuroendocrine tumour liver metastases: comparing conventional TACE, drug-eluting beads TACE and yttrium-90 radioembolisation as treatment options using a propensity score analysis model. Eur Radiol 2017;27:4995-5005. [Crossref] [PubMed]
- Clift AK, Frilling A. Liver transplantation and multivisceral transplantation in the management of patients with advanced neuroendocrine tumours. World J Gastroenterol 2018;24:2152-62. [Crossref] [PubMed]
- Spolverato G, Bagante F, Tsilimigras DI, et al. Liver transplantation in patients with liver metastases from neuroendocrine tumors. Minerva Chir 2019;74:399-406. [Crossref] [PubMed]
- Mazzaferro V, Pulvirenti A, Coppa J. Neuroendocrine tumors metastatic to the liver: how to select patients for liver transplantation? J Hepatol 2007;47:460-6. [Crossref] [PubMed]
- Cloyd JM, Konda B, Shah MH, et al. The emerging role of targeted therapies for advanced well-differentiated gastroenteropancreatic neuroendocrine tumors. Expert Rev Clin Pharmacol 2019;12:101-8. [Crossref] [PubMed]
- Rinke A, Müller HH, Schade-Brittinger C, et al. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol 2009;27:4656-63. [Crossref] [PubMed]
- Moertel CG, Hanley JA, Johnson LA. Streptozocin alone compared with streptozocin plus fluorouracil in the treatment of advanced islet-cell carcinoma. N Engl J Med 1980;303:1189-94. [Crossref] [PubMed]
- Kunz P. A randomized study of temozolomide or temozolomide and capecitabine in patients with advanced pancreatic neuroendocrine tumors: A trial of the ECOG-ACRIN Cancer Research Group (E2211). J Clin Oncol 2018;36:abstr 4004.


