
Treatment delays during FOLFOX chemotherapy in patients with colorectal cancer: a multicenter retrospective analysis
Introduction
Delays in chemotherapy administration are common and can have disruptive effects on patients’ experiences during cancer treatment (1,2). Some treatment delays are elective, such as delays for holidays or personal obligations; other delays are obligatory, such as interruptions for acute infection or other illness. Still other delays occur at the discretion of the managing clinician, such as when patients present for treatment with low blood cell counts. This last type of delay can place a substantial burden on patients and their caregivers by causing them to travel to the treatment center, take time off from work, and spend additional time away from their families—all without the benefit of receiving the planned chemotherapy treatment. These delays are often preceded by a trend of declining blood count values, raising the possibility that they may be predictable. Furthermore, these discretionary delays lead to undesirable health system inefficiencies, leaving unused space in the infusion suite at the time of the delay while incurring additional out-of-pocket costs and future clinical encounters for patients.
Chemotherapy treatment delays are common in many cancer treatment settings and with many different chemotherapy regimens. Delays can have differing implications depending on the goals of treatment (e.g., adjuvant vs. palliative-intent therapy) and the feasibility of altering the treatment plan to prevent a delay. We chose to study chemotherapy treatment delays among colorectal cancer patients receiving FOLFOX chemotherapy (folinic acid, 5-fluorouracil, and oxaliplatin) because this is a widely-used treatment regimen during which delays and dose-limiting toxicities are common (1-5).
In clinical experience, delays during FOLFOX are often caused by neutropenia. Clinical trial protocols for delivery of FOLFOX chemotherapy have generally mandated delaying treatment for patients presenting with an absolute neutrophil count (ANC) that is less than either 1,500 or 1,000 neutrophils/mm3, and for platelet counts less than 100,000 or 50,000 platelets/mm3 (6,7). The necessity of delaying chemotherapy treatment when blood counts are below these thresholds is uncertain, especially at their upper ranges. Chemotherapy is often withheld out of concern for febrile neutropenia, however existing reports suggest that this complication is very infrequent with FOLFOX chemotherapy (reported incidence of 0–8%) (1,4,6,7). In routine clinical practice there are no established recommendations for when to delay treatment or adjust dosing, and the decision to delay chemotherapy treatment is provider-dependent.
The effects of chemotherapy delays and dose modifications on colorectal cancer treatment outcomes are not well understood. However, the costs and inefficiencies associated with chemotherapy treatment delays are frequently evident to us in clinical practice. We conducted this study to better understand the frequency and causes of treatment delays during FOLFOX chemotherapy.
Methods
Study design and patient selection
We performed a retrospective cohort study of patients receiving FOLFOX chemotherapy for treatment of colorectal cancer. Using electronic chemotherapy treatment records, we identified patients who received FOLFOX-type regimens between January 2012 and April 2016 at either the Norris Cotton Cancer Center (Lebanon, New Hampshire) or the University of Colorado Cancer Center (Aurora, Colorado). We defined FOLFOX-type regimens as chemotherapy regimens containing oxaliplatin and infusional 5-FU, at a minimum, on a standard 14-day treatment cycle. We included patients who received concurrent bevacizumab, cetuximab, or panitumumab. Both institutions used mFOLFOX6 as the standard FOLFOX protocol during this period, though leucovorin doses were sometimes reduced or omitted due to drug shortages. Patients receiving qualifying chemotherapy regimens were included regardless of cancer stage or therapeutic intent. Patients who were treated as part of clinical trials were excluded, as we aimed to study treatment delays in routine clinical care. The study was reviewed and approved by the institutional review board at each of the two participating centers.
Outcomes
The primary outcome measure was the occurrence of unplanned chemotherapy treatment delays. Delays were assessed in cycles 2 through 6; we did not attempt to assess delays in chemotherapy initiation (cycle 1 delays), and we limited our analysis to the first six cycles of chemotherapy because we expected that duration of therapy would vary substantially after completion of six treatment cycles. We defined a treatment delay as an interval of more than 18 days between chemotherapy treatments, with the expectation that most non-delayed cycles would have 14-day intervals. Delays were considered unplanned when the delay was not anticipated by the treating oncologist at the start of the preceding treatment cycle. For example, delays due to patient travel or holiday plans or planned surgical procedures were classified as planned delays, and were not counted under the primary study outcome. We also did not consider chemotherapy cycles to be delayed when treatment was discontinued or changed due to disease progression.
Data collection
One reviewer abstracted data from the electronic medical record at each site. Baseline data collection included patient age, sex, and therapeutic intent (curative/adjuvant vs. other, as categorized by the treating physician). When treatments were found to be delayed, the reason for delay was determined from review of the treating provider’s documentation in the medical record (including whether the delay was planned or unplanned). In instances where the rationale for delay was not explicitly documented, we reviewed laboratory data to identify the apparent reason for delay. Reasons for delay were recorded in qualitative notes, then categorized collaboratively by two or more members of the research team.
Analysis
We summarized our findings descriptively. The main outcome measures were the proportion of all evaluated chemotherapy cycles with unplanned delays, the proportion of patients with at least one unplanned delay (cycles 2–6), and the primary reasons for unplanned treatment delays. In addition to presenting the findings for the overall population, we performed analyses stratified by age group (categorized as younger than 55, 55–64, 65–74, and 75 years and older) and therapeutic intent (adjuvant/curative-intent vs. palliative/other). We used Chi-square tests to evaluate for categorical between-group differences in unplanned delays; we considered findings to be statistically significant at a threshold of P<0.05.
Results
Across the two study sites we identified 214 patients receiving standard-of-care FOLFOX chemotherapy for colorectal cancer. The median age was 59 years, 55% were female, and 58% were receiving curative-intent (i.e., adjuvant) therapy. Cycle 1 dose reductions of one or more chemotherapy agents were used in 27 patients (13%). Additional patient clinical and demographic information is presented in Table 1.

Full table
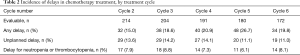
We identified 961 chemotherapy cycles that were evaluable for delays (maximum of five evaluable cycles per patient). Unplanned delays occurred in 124 cycles (13%), and 43% of patients had at least one unplanned delay prior to receipt of the sixth treatment cycle. Planned delays occurred in an additional 68 cycles (7%). Table 2 shows the incidence of delays by cycle; unplanned delays occurred with similar frequency across cycles 2 through 6.
Full table
The most common reasons for unplanned treatment delays were neutropenia (accounting for 44% of unplanned delays) and thrombocytopenia (16%). Additional reasons for unplanned delays are shown in Table 3. Seventy-two of 214 patients (34%) had at least one delay for neutropenia and/or thrombocytopenia during cycles 2–6. Among patients with delays caused by cytopenias there was a wide range in cell counts at the time of delay. In cycles delayed for neutropenia, ANC ranged from 0.18 to 1.36 K/mm3 [median 0.89 K/mm3, interquartile range (IQR), 0.70–1.10]. In cycles delayed for thrombocytopenia, platelet counts at the time of the delay ranged from 52 to 97 K/mm3 (median 76.0 K/mm3, IQR, 63–85). We identified only one documented episode of febrile neutropenia in this analysis.

Full table
We performed subgroup analyses with stratification by age group and by curative-intent vs. palliative chemotherapy. There was no significant difference in the incidence of unplanned treatment delays by age group (Chi square P value =0.54), with one or more unplanned delays occurring in 38% of patients age 54 years and younger (n=85), 49% of patients age 55–64 (n=65), 42% of patients age 65–74 (n=45), and 47% of patients age 75 and older (n=19). The incidence of unplanned delays also did not differ significantly between patients receiving curative-intent vs. palliative treatment (44% vs. 41% of patients with unplanned delays, P=0.60). Of note, the proportions of unplanned delays attributable to hematologic toxicities was greater in curative-intent vs. palliative treatment (67% vs. 45% of cycles, P=0.03).
Discussion
The main objective of our study was to determine the frequency of unplanned delays in patients receiving FOLFOX chemotherapy. Unplanned delays affected 13% of all evaluated chemotherapy cycles, and almost half of our study population (43%) experienced at least one unplanned delay during the first six cycles of treatment. Cytopenias, primarily neutropenia, were the main contributing cause for delay in 60% of cycles with unplanned delays. Cytopenias were rarely severe in degree, and we identified only one episode of fever and neutropenia in our cohort of 214 patients. These findings were generalizable across patients receiving chemotherapy with either adjuvant or palliative intent.
Our findings regarding the prevalence and causes of unplanned delays are largely consistent with prior studies of real-world populations. Smoragiewicz et al. studied 114 patients receiving FOLFOX as adjuvant chemotherapy for colon cancer; they observed dose-limiting toxicities in 22% of treatment cycles, and most dose-limiting toxicities were associated with treatment delays (1). Neutropenia accounted for 51% of all dose-limiting toxicities in this series. Kim et al. reported on 246 patients receiving adjuvant chemotherapy for colon cancer, primarily with FOLFOX (2). After three months of treatment (approximately six treatment cycles) 30% of patients had experienced a treatment delay, with hematologic toxicities accounting for most delays.
In a notable divergence from our own study and from those described above, Chiarotto and Dranitsaris report very low rates of chemotherapy treatment delays during FOLFOX chemotherapy (2.2% of all chemotherapy cycles) (8). Typical thresholds for neutrophil or platelet counts were not applied in this observational series, and treatment was delivered without delay in patients with neutrophil counts as low as 100 neutrophils/mm3 (with dose-reductions in some cases). Despite this aggressive approach, febrile neutropenia was observed in only six patients (4.6%) in this cohort. In summary, the existing literature suggests that delays and “dose-limiting” toxicities are frequent in routine practice; however, there is scant evidence that delaying chemotherapy using typical blood count thresholds is necessary to preserve patient safety.
A key implication of our findings is that many cytopenia-related delays during FOLFOX chemotherapy may be unnecessary. Strategies for avoiding these cytopenia-related delays could include implementation of revised default criteria for neutrophil and platelet count treatment thresholds (9,10). The experience reported by Chiarotto and Dranitsaris (8) suggests that commonly employed ANC thresholds are highly conservative (e.g., delaying chemotherapy for ANC <1 or 1.5 K/mm3), and that rates of febrile neutropenia are low even when FOLFOX is given at much lower neutrophil counts. Since current treatment strategies commonly employ both a delay and a chemotherapy dose reduction for cytopenias, an alternative approach could be to implement dose reductions instead of delaying treatment. The safety of strategies for avoiding cytopenia-related treatment delays could be further supported by implementation of pretreatment pharmacogenomic testing to identify uncommon DPYD gene polymorphisms that are associated with severe and potentially life-threatening neutropenia during fluoropyrimidine chemotherapy (11,12). While pharmacogenetic screening for DPYD mutations has not yet been adopted at scale or included in colorectal cancer treatment guidelines, substantial new evidence to support this practice has emerged in recent years (13,14).
An additional strategy for avoiding neutropenia-related delays could be to increase the use of granulocyte colony stimulating factor (G-CSF). Prior studies have demonstrated that use of G-CSF with chemotherapy can reduce the incidence of neutropenia, febrile neutropenia, and chemotherapy delays during palliative treatment of colorectal cancer (15,16). These studies included twice-daily temperature measurement, which is not standard practice during chemotherapy treatment and which may have led to overdetection of febrile episodes. In the case of colorectal cancer, evidence is lacking to support an association of G-CSF use with improved overall survival in either the adjuvant or palliative setting. Furthermore, pegylated G-CSF agents are costly (average sales price of $4,681 per administration) (17), and patient copays for these drugs can contribute to the increasingly recognized problem of “financial toxicity” (18). For these reasons, we do not recommend strategies that would involve reflexive use of G-CSF in order to reduce chemotherapy treatment delays.
Some limitations of our study include the relatively small sample size and the retrospective nature of the study. However, our findings are consistent with prior research. Our focus on delays in chemotherapy treatment is a strength of this study, as prior studies have focused on chemotherapy dose-intensity or dose-limiting toxicities without a specific focus on treatment delays.
In summary, unplanned delays are common during treatment with FOLFOX chemotherapy, and most delays are attributable to hematologic toxicities (especially neutropenia). At the same time, thresholds for delaying chemotherapy due to neutropenia or thrombocytopenia are not evidence-based. Delays in chemotherapy treatment reduce chemotherapy dose-intensity, and unplanned delays lead to more office visits and increased out-of-pocket costs for patients. Strategies for reducing unplanned delays include relaxation of overly stringent hematologic treatment criteria, use of selective chemotherapy dose adjustments without concomitant delay, and use of G-CSF without concomitant delay (if the alternative strategy would have been G-CSF use after a delay). We suggest the development and testing of well-defined, proactive approaches using these strategies during FOLFOX chemotherapy treatment, particularly in the case of otherwise asymptomatic grade 3 neutropenia (ANC 500–999/mm3).
Acknowledgments
This work was supported by the Conquer Cancer Foundation (Career Development Award to GA Brooks).
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was reviewed and approved by the institutional review board at each of the two participating centers.
References
- Smoragiewicz M, Javaheri KR, Yin Y, et al. Neutropenia and relative dose intensity on adjuvant FOLFOX chemotherapy are not associated with survival for resected colon cancer. J Gastrointest Cancer 2014;45:460-5. [Crossref] [PubMed]
- Kim JH, Baek MJ, Ahn BK, et al. Clinical Practice in the Use of Adjuvant Chemotherapy for Patients with Colon Cancer in South Korea: a Multi-Center, Prospective, Observational Study. J Cancer 2016;7:136-43. [Crossref] [PubMed]
- Satkunam N, Wei X, Biagi JJ, et al. Delivery of Adjuvant Oxaliplatin for Colon Cancer: Insights From Routine Clinical Practice. J Natl Compr Canc Netw 2016;14:1548-54. [Crossref] [PubMed]
- Uncu D, Aksoy S, Cetin B, et al. Results of adjuvant FOLFOX regimens in stage III colorectal cancer patients: retrospective analysis of 667 patients. Oncology 2013;84:240-5. [Crossref] [PubMed]
- Cespedes Feliciano EM, Lee VS, Prado CM, et al. Muscle mass at the time of diagnosis of nonmetastatic colon cancer and early discontinuation of chemotherapy, delays, and dose reductions on adjuvant FOLFOX: The C-SCANS study. Cancer 2017;123:4868-77. [Crossref] [PubMed]
- Andre T, Boni C, Mounedji-Boudiaf L, et al. Oxaliplatin, fluorouracil, and leucovorin as adjuvant treatment for colon cancer. N Engl J Med 2004;350:2343-51. [Crossref] [PubMed]
- Allegra CJ, Yothers G, O'Connell MJ, et al. Initial safety report of NSABP C-08: A randomized phase III study of modified FOLFOX6 with or without bevacizumab for the adjuvant treatment of patients with stage II or III colon cancer. J Clin Oncol 2009;27:3385-90. [Crossref] [PubMed]
- Chiarotto JA, Dranitsaris G. FOLFOX chemotherapy can safely be given to neutropenic patients with early-stage colorectal cancer for higher dose intensity and fewer visits. Support Care Cancer 2016;24:2533-9. [Crossref] [PubMed]
- Ojerholm E, Halpern SD, Bekelman JE. Default Options: Opportunities to Improve Quality and Value in Oncology. J Clin Oncol 2016;34:1844-7. [Crossref] [PubMed]
- Yoshida Y, Hoshino S, Aisu N, et al. Can grade 2 neutropenia predict the risk of grade 3 neutropenia in metastatic colorectal cancer patients treated with chemotherapy? Support Care Cancer 2015;23:1623-7. [Crossref] [PubMed]
- Lunenburg CATC, Henricks LM, Guchelaar HJ, et al. Prospective DPYD genotyping to reduce the risk of fluoropyrimidine-induced severe toxicity: Ready for prime time. Eur J Cancer 2016;54:40-8. [Crossref] [PubMed]
- Henricks LM, Opdam FL, Beijnen JH, et al. DPYD genotype-guided dose individualization to improve patient safety of fluoropyrimidine therapy: call for a drug label update. Ann Oncol 2017;28:2915-22. [Crossref] [PubMed]
- Deenen MJ, Meulendijks D, Cats A, et al. Upfront Genotyping of DPYD*2A to Individualize Fluoropyrimidine Therapy: A Safety and Cost Analysis. J Clin Oncol 2016;34:227-34. [Crossref] [PubMed]
- Henricks LM, Lunenburg C, de Man FM, et al. DPYD genotype-guided dose individualisation of fluoropyrimidine therapy in patients with cancer: a prospective safety analysis. Lancet Oncol 2018;19:1459-67. [Crossref] [PubMed]
- Hecht JR, Pillai M, Gollard R, et al. A randomized, placebo-controlled phase II study evaluating the reduction of neutropenia and febrile neutropenia in patients with colorectal cancer receiving pegfilgrastim with every-2-week chemotherapy. Clin Colorectal Cancer 2010;9:95-101. [Crossref] [PubMed]
- Pinter T, Klippel Z, Cesas A, et al. A Phase III, Randomized, Double-Blind, Placebo-Controlled Trial of Pegfilgrastim in Patients Receiving First-Line FOLFOX/Bevacizumab or FOLFIRI/Bevacizumab for Locally Advanced or Metastatic Colorectal Cancer: Final Results of the Pegfilgrastim and Anti-VEGF Evaluation Study (PAVES). Clin Colorectal Cancer 2017;16:103-14.e3. [Crossref] [PubMed]
- Center for Medicare and Medicaid Services. Medicare Part B Drug Average Sales Price > 2019 ASP Drug Pricing Files. Available online: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Part-B-Drugs/McrPartBDrugAvgSalesPrice/2019ASPFiles.html. Accessed January 22, 2019.
- Zafar SY, Peppercorn JM, Schrag D, et al. The financial toxicity of cancer treatment: a pilot study assessing out-of-pocket expenses and the insured cancer patient's experience. Oncologist 2013;18:381-90. [Crossref] [PubMed]


