
Transanal total mesorectal excision for rectal cancer: hype or new hope?
Background
Rectal cancer has always posed surgical challenges to the colorectal surgeon—confinement of the lesion of interest within the pelvis poses spatial limitations and hinders ideal tumor resection, leading to the possibility of local recurrence and subsequent poor oncological and survival outcomes.
In 1982, Dr. Heald et al. proposed total mesorectal excision (TME), a surgical advancement technique emphasizing sharp, nerve-sparing circumferential dissection along the congenital avascular plane between the mesorectum and surrounding tissue (1). By not disrupting the mesorectal fascia, TME dramatically decreased local recurrence of rectal cancer and has since then become the surgical gold standard for curative resection. Furthermore, the aid of neoadjuvant/adjuvant chemotherapy and radiotherapy has even further decreased local recurrence rates.
It comes, as no surprise then, that the quality of the TME performed is key in determining local disease control. Studies have demonstrated that poor surgical TME quality is directly linked to local malignancy recurrence that unfortunately cannot be rectified by chemoradiotherapy. Recurrence is notoriously difficult to treat and not only immensely impacts quality of life, but also imbues a worse prognosis going forward (2).
The 1990’s saw the introduction of laparoscopic surgery and its eventual role in colorectal surgery. While there were initial concerns regarding laparoscopy’s oncological safety, clinical trials including outcomes of surgical therapy like COST (3) and COLOR (4) subsequently demonstrated the safety and clinical benefit noninferiority of laparoscopy to traditional open colon surgery. Unfortunately, studies comparing laparoscopy to open rectal surgery including COLOR II (5), ALaCaRT (6), and ACOSOG Z6051 (7,8), all revealed noninferiority was not achieved. The investigators of these rectal surgery studies pointed towards numerous factors—the complexity of traction and counter-traction while utilizing a straight rigid instrument, limited visualization offered via laparoscopy in a restricted operative space, and imperfections of linear stapling in transecting the rectum—as reasons why laparoscopy, unless in the most experienced of surgical hands, most likely still yielded similar outcomes to open surgery. With such a subjective variability involved, it thus became difficult to generalize laparoscopy’s use in treating rectal cancer.
Robotic surgery then arose in response to the aforementioned limitations by attempting to offer better visualization and increased instrument articulation. While there were anecdotal reports that robotics did indeed lead to better TME quality, lower circumferential resection margin (CRM) positivity, and lower conversion rate, the recently published ROLLAR trial did not support these proposed advances over traditional laparoscopy (9). In fact, only one sub-group analysis of higher body mass index (BMI) male patients supported the superiority of robotics. In addition, one must not forget the issue of cost-effectiveness of robotic surgery.
As a result, in the current state of affairs where the advancement and cost of robotics will determine robotic surgery’s fate, alongside evidence demonstrating that open surgery still reigns supreme as the gold standard in surgical treatment of rectal cancer, colorectal surgeons are left searching for the next breakthrough in surgical treatment of rectal cancer.
The evolution of transanal TME (taTME)
The year 1983 saw the development of transanal endoscopic microsurgery (TEM) by Buess et al. by facilitating magnified visualization during transanal resection (10), TEM not only allowed greater precision in rectal lesion excision when compared to during traditional open surgical approaches, but also yielded lower morbidity, lower local recurrence rates, and higher rates of negative resection margins. However, TEM never truly became a standard, owing to the cost of the specialized surgical instrument needed, as well as the procedure’s limited indications and steep learning curve. Nevertheless, it still firmly rooted the concept of integrating laparoscopy via natural orifices into the general approach of minimally invasive surgery.
Albert et al. subsequently proposed Transanal Minimally Invasive Surgery (TAMIS) using an advanced transanal platform developed by GelPOINT Path® (Applied Medical, USA) (11). This platform quickly propelled TAMIS to much greater heights than TEM, given that TAMIS offered the familiarity of standard laparoscopic instrumentation combined with a superior visualization field that did not require purchase of additional specialized devices.
The latest advancement in this rapidly progressing field has emerged to be NOTES—Natural Orifice Transluminal Endoscopic Surgery. After procedures including cholecystectomy, appendectomy, and even rectosigmoid resection were completed via NOTES in both animal models and clinical patients, 2010 saw the first series of 20 patients who underwent taTME by de Lacy et al. This team was able to successfully perform the most difficult portion of rectal surgery—TME—via a novel “bottom up” approach (12). Many subsequent studies have demonstrated similar successes, proving the safety and feasibility of this innovative method (13). Heald et al. had hailed taTME as “a new solution to some old problems” and predicted 2013 to be the year of taTME (14).
As a result, taTME is not a novel concept. Rather, it is a culmination of inspiration and innovation founded in the advancements offered by TEM, TAMIS, and NOTES.
The evolution of surgical technique
The development of taTME was motivated by difficulties surrounding lower rectal dissection. The transanal approach offered a shorter distance to approach most difficult location from below, and the in-line positioning was ideal in providing optimal visualization of the operative field to determine adequate distal resection margin and thereby increase the chances of preserving the sphincter. Early taTME studies dwelled on debates regarding transanal platform choice, surgical approach and technique, and final anastomosis creation method (13). Most innovative is the fact that taTME allows for two surgical teams to simultaneously operate on one case—Ceceil approach. This approach decreases operative difficulty, as well as operative time by approximately sixty minutes (15).
At its current stage of development, taTME has become standardized in its surgical techniques and identification of anatomical landmarks via the transanal view, thereby facilitating its reproducibility in ongoing cases. However, final anastomosis creation method remains a somewhat work in progress. In a preliminary series, over 60% of anastomoses were hand-sewn (12), but more recent series have yielded results as far of 40% hand-sewn versus 60% circular stapler performed (16). These latest statistics mirror those of traditional laparoscopic and robotic surgery and indirectly support the notion that distance of the tumor from the sphincter, rather than surgical technique, should determine method of anastomosis.
Complete taTME without transabdominal assistance—otherwise known as NOTES (Natural Orifice Tele-Endoscopic Surgery)—was once hypothesized to be the ultimate evolution of taTME. Case reports elicited difficulties arising from severely limited patient eligibility criteria and lack of appropriate surgical instrumentation (17,18). As a result, taTME has not yet progressed towards NOTES at this time.
taTME indications
The St. Gallen Consensus concluded that taTME may be technically easier than traditional abdominal TME in patients with narrow pelvic anatomy, obesity, and bulky mid-to-distal rectal tumors (19). However, if one considers usual markers for a so-called “difficult” pelvis—BMI and hip-waist ratio—there are no pre-determined cutoffs to help surgeons in deciding traditional TME versus taTME (19).
As touched upon earlier in this review, taTME’s innovative “bottom up” approach lessens the technical limitations present in laparoscopic TME and facilitates increased operative completeness and sufficiency. Congruently, a meta-analysis completed by Ma et al. demonstrated that laparoscopic TME carries a 4-time higher likelihood of conversion when compared to taTME (20). In addition, taTME also facilitates higher rates of complete TME quality, lower rates of CRM positivity, and increased CRM length (21). This superiority of pathological results may become the strongest motivator for performing taTME; however, further investigations are definitely necessary before this conclusion can be definitively drawn. Furthermore, the two-team simultaneous surgical approach can effectively shorten operative time by up to 60 minutes, indirectly suggesting that postoperative recovery can also be expedited. However, the resources involved in such an endeavor, both personnel and equipment wise, is not generalizable to all medical facilities.
Lastly, taTME has been extended to treatment of other disease pathologies as well. These include Crohn’s disease, ulcerative colitis, revision of anastomosis strictures, and reversal of Hartmann’s procedure.
Complications
With any new and technically challenging surgical approach, safety remains paramount over efficacy. Reported 30-day taTME-associated morbidity varies widely from 8.7% to 52%, while short-term taTME morbidity averages 31.5% in comparison to 39.6% for laparoscopic TME (21).
Urethral injury
Injury to the membranous portion of the urethra during taTME often arises secondary to unfamiliarity of the dissection plane anterior to the rectum in male patients. The international taTME registry currently documents a 0.8% risk of urethral injury amongst a database of 720 patients (22). Consequences of this complication are particularly difficult to address, especially in those who have already received neoadjuvant radiotherapy and are left with impaired tissue healing abilities. This specific complication can be mitigated via structured taTME training followed by mentored operative supervision.
Pelvic abscess
Since part of taTME occurs while the rectal lumen remains open, pelvic contamination and subsequent abscess formation has been reported to be as a high as 16.2% (23). With improvement and standardization of surgical protocol, rates have improved to 2.3–2.6% (24). Subsequent studies have suggested that anastomotic complication, rather than the taTME procedure itself, is the root cause (15,25).
Pelvic wall bleeding
Excessive lateral pelvic wall dissection can lead to bleeding, most often arising from lateral pelvic side-wall vessels surrounding the mid-rectum. The aforementioned taTME registry currently documents an incidence rate of 6.9% (22). Insufflating the pelvis, particularly with use of advanced insufflators such as AirSeal® (ConMed Corporation, Milford, CT, USA), may mitigate some of the risk, but can also create false areolar planes and mislead the surgeon. Massive intraoperative bleeding will most definitely call for open surgical conversion.
Anastomotic leak
Anastomotic leak is considered the major complication in lower rectal surgery. The double stapling technique remains the primary anastomosis method in both laparoscopic and robotic transabdominal approaches, and the difficulties associated with applying a linear stapler in the pelvis’ narrow operative field and having to fire multiple times in order to transect the rectum greatly raise the chance of resultant anastomotic leak (26).
Given that taTME utilizes a single circular stapler in 70–75% of cases, proponents of taTME strongly believe that the single stapler method leads to decreased likelihood of anastomotic leak. Unfortunately, data thus far paints a mixed picture. The aforementioned taTME registry documents an anastomotic leak rate of 15.7% with 79% of those cases requiring repeat intervention (16), while the also aforementioned analysis by Ma et al. documents a leak rate of 10% (20). Another recently published multicenter case-matched study documents a rate of 11.1% as compared to a rate of 9.5% in robotic cases, but failed to reach statistical significance upon analysis (27). In a multicenter prospective audit of elective rectal cancer surgeries from the European Society of Coloproctology (ESCP) collaborating group, laparoscopic taTME [odds ratio (OR) 1.61, P=0.04] and robotic taTME (OR 3.05, P=0.02) were both associated with higher risks of anastomotic leak when compared to non-transanal laparoscopic or robotic TME. However, this association was subsequently lost when a mixed effect model controlling for patient and disease factors was applied (28).
Thus, literature has failed to support the initial notion that single stapling would decrease rates of anastomotic leak. However, one must keep in mind that taTME, particularly robotic-assisted taTME, has come a long way since the initial times these data arise from, and one wonders whether technique maturation will lead to fruition of the initial notion. Nevertheless, taTME remains technically challenging and requires intensive multimodal training—lectures, hands-on cadaver training, proctored clinical application—in order to protect patients from reckless employment of the technique and subsequent devastating complications. Most importantly, one must refer to the St. Gallen consensus for safe implementation of the technique (19).
Functional results
Bowel, bladder, and sexual dysfunction rank amongst the most common and devastating postoperative functional impairments arising secondary to rectal cancer surgery. While taTME may increase the chances of performing sphincter-preserving surgery, the lower anatomical anastomosis poses a counterpoint and may in fact cause greater impairments in anal function. This risk is further increased by the transanal platform intraoperatively stretching the anal sphincter. As for urinary and sexual function, the taTME approach provides better visualization of the operative field, facilitating neurovascular bundle preservation and thus increasing the chances and degree of preserving urinary and sexual function.
A recent series report encompassing 30 patients’ status post taTME at 6 months revealed noninferior quality of life and functional outcomes when compared to those who underwent conventional laparoscopic low anterior resection (29). However, it must be noted that at the one-month postoperative point, all evaluated markers actually demonstrated a decline with the majority improving back to comparable baseline at the six-month mark—except for anal pain and social functioning. In another study comparing 27 patients undergoing laparoscopic TME to another 27 undergoing taTME, low anterior resection syndrome (LARS) at 6 months post stoma reversal was higher in the taTME group (16/27 versus 8/27), but failed to elicit statistical significance. As a result, the authors concluded that functional outcomes and quality of life outcomes were similar between the two groups (30).
Therefore, at this point in time, it is simply too early to draw any preliminary conclusions regarding taTME functional results.
Oncologic results
taTME was developed on the premise that it could achieve higher quality TME. The aforementioned international taTME registry currently demonstrates a near-complete to complete mesorectal excision rate of 96%, CRM positivity rate of 2.4%, and distal rectal margin (DRM) positivity rate of 0.3% (22). In our own case-matched study, taTME yielded statistically significant longer distal margin lengths in both middle and lower rectal cancer surgeries. Furthermore, taTME also yielded longer CRM distances and a less than 1mm incidence rate (4% versus 10%) (15). The previously reference meta-analysis from Ma et al. similarly concluded that taTME is able to obtain significantly higher rates of near-complete to complete mesorectal excision when compared to that of laparoscopic TME, and that taTME also yields longer CRM distances with a significantly lower risk of CRM positivity (20).
While these short-term pathological advantages have been re-created in numerous small preliminary taTME reports, whether or not these advantages in fact translate into the ultimate goals of lower local recurrence rate and prolonged long-term survival remains in question. Lelong et al. demonstrated a 5.3% versus 5.7% local recurrence rate in laparoscopic versus taTME groups, respectively, at 31.9-month follow-up (31). While this result seems somewhat disappointing, one must keep in mind that only 72 patients were included in this study, thereby highlighting the need for multicenter, long-term, and large randomized controlled trials for taTME to truly evaluate long-term oncologic outcomes.
Ongoing clinical investigations
A quick search on clinicaltrials.gov reveals over ten randomized clinical trials evaluating laparoscopic TME versus taTME. While the majority do not offer much public detail, Table 1 summarized two well-known and actively ongoing studies of interest.
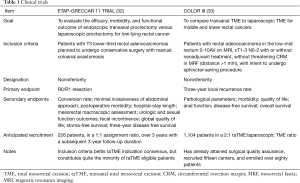
Full table
Conclusions
taTME, while definitely still in its early stages of development, has steadily accumulated safety and feasibility data while simultaneously adapting to said data in order to grow and mature. It not only provides a better solution to an old problem that colorectal surgeons have been attempting to tackle for quite some time, but also appears to be quite promising in terms of outcomes on numerous fronts. As mentioned earlier, with intensive multimodal training that must include lectures, hands-on cadaver training, and proctored clinical application, alongside design and implementation of international-scale large multicenter randomized clinical trials, one can only hope that taTME and its innovations will not only open a new era for colorectal surgery, but also for even more surgical disease pathologies.
Acknowledgments
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
References
- Heald RJ, Husband EM, Ryall RD. The mesorectum in rectal cancer surgery--the clue to pelvic recurrence? Br J Surg 1982;69:613-6. [Crossref] [PubMed]
- Quirke P, Durdey P, Dixon MF, et al. Local recurrence of rectal adenocarcinoma due to inadequate surgical resection. Histopathological study of lateral tumour spread and surgical excision. Lancet 1986;2:996-9. [Crossref] [PubMed]
- Clinical Outcomes of Surgical Therapy Study Group, Nelson H, Sargent DJ, et al. A comparison of laparoscopically assisted and open colectomy for colon cancer. N Engl J Med 2004;350:2050-9. [Crossref] [PubMed]
- Veldkamp R, Kuhry E, Hop WC, et al. Laparoscopic surgery versus open surgery for colon cancer: short-term outcomes of a randomised trial. Lancet Oncol 2005;6:477-84. [Crossref] [PubMed]
- Bonjer HJ, Deijen CL, Abis GA, et al. A randomized trial of laparoscopic versus open surgery for rectal cancer. N Engl J Med 2015;372:1324-32. [Crossref] [PubMed]
- Stevenson AR, Solomon MJ, Lumley JW, et al. Effect of Laparoscopic-Assisted Resection vs Open Resection on Pathological Outcomes in Rectal Cancer: The ALaCaRT Randomized Clinical Trial. JAMA 2015;314:1356-63. [Crossref] [PubMed]
- Fleshman J, Branda ME, Sargent DJ, et al. Disease-free Survival and Local Recurrence for Laparoscopic Resection Compared With Open Resection of Stage II to III Rectal Cancer: Follow-up Results of the ACOSOG Z6051 Randomized Controlled Trial. Ann Surg 2019;269:589-95. [Crossref] [PubMed]
- Fleshman J, Branda M, Sargent DJ, et al. Effect of Laparoscopic-Assisted Resection vs Open Resection of Stage II or III Rectal Cancer on Pathologic Outcomes: The ACOSOG Z6051 Randomized Clinical Trial. JAMA 2015;314:1346-55. [Crossref] [PubMed]
- Jayne D, Pigazzi A, Marshall H, et al. Effect of Robotic-Assisted vs Conventional Laparoscopic Surgery on Risk of Conversion to Open Laparotomy Among Patients Undergoing Resection for Rectal Cancer: The ROLARR Randomized Clinical Trial. JAMA 2017;318:1569-80. [Crossref] [PubMed]
- Raestrup H, Manncke K, Mentges B, et al. Indications and technique for TEM (transanal endoscopic microsurgery). Endosc Surg Allied Technol 1994;2:241-6. [PubMed]
- Albert MR, Atallah SB, deBeche-Adams TC, et al. Transanal minimally invasive surgery (TAMIS) for local excision of benign neoplasms and early-stage rectal cancer: efficacy and outcomes in the first 50 patients. Dis Colon Rectum 2013;56:301-7. [Crossref] [PubMed]
- de Lacy AM, Rattner DW, Adelsdorfer C, et al. Transanal natural orifice transluminal endoscopic surgery (NOTES) rectal resection: "down-to-up" total mesorectal excision (TME)--short-term outcomes in the first 20 cases. Surg Endosc 2013;27:3165-72. [Crossref] [PubMed]
- Chen CC, Lai YL, Jiang JK, et al. The evolving practice of hybrid natural orifice transluminal endoscopic surgery (NOTES) for rectal cancer. Surg Endosc 2015;29:119-26. [Crossref] [PubMed]
- Heald RJ. A new solution to some old problems: transanal TME. Tech Coloproctol 2013;17:257-8. [Crossref] [PubMed]
- Chen CC, Lai YL, Jiang JK, et al. Transanal Total Mesorectal Excision Versus Laparoscopic Surgery for Rectal Cancer Receiving Neoadjuvant Chemoradiation: A Matched Case-Control Study. Ann Surg Oncol 2016;23:1169-76. [Crossref] [PubMed]
- Penna M, Hompes R, Arnold S, et al. Incidence and Risk Factors for Anastomotic Failure in 1594 Patients Treated by Transanal Total Mesorectal Excision: Results From the International TaTME Registry. Ann Surg 2019;269:700-11. [Crossref] [PubMed]
- Zhang H, Zhang YS, Jin XW, et al. Transanal single-port laparoscopic total mesorectal excision in the treatment of rectal cancer. Tech Coloproctol 2013;17:117-23. [Crossref] [PubMed]
- Kang L, Chen WH, Luo SL, et al. Transanal total mesorectal excision for rectal cancer: a preliminary report. Surg Endosc 2016;30:2552-62. [Crossref] [PubMed]
- Adamina M, Buchs NC, Penna M, et al. St.Gallen Colorectal Consensus Expert G: St.Gallen consensus on safe implementation of transanal total mesorectal excision. Surg Endosc 2018;32:1091-103. [Crossref] [PubMed]
- Ma B, Gao P, Song Y, et al. Transanal total mesorectal excision (taTME) for rectal cancer: a systematic review and meta-analysis of oncological and perioperative outcomes compared with laparoscopic total mesorectal excision. BMC Cancer 2016;16:380. [Crossref] [PubMed]
- Emile SH, de Lacy FB, Keller DS, et al. Evolution of transanal total mesorectal excision for rectal cancer: From top to bottom. World J Gastrointest Surg 2018;10:28-39. [Crossref] [PubMed]
- Penna M, Hompes R, Arnold S, et al. Transanal Total Mesorectal Excision: International Registry Results of the First 720 Cases. Ann Surg 2017;266:111-7. [Crossref] [PubMed]
- Velthuis S, Veltcamp Helbach M, Tuynman JB, et al. Intra-abdominal bacterial contamination in TAMIS total mesorectal excision for rectal carcinoma: a prospective study. Surg Endosc 2015;29:3319-23. [Crossref] [PubMed]
- van Oostendorp SE, Koedam TW, Sietses C, et al. Transanal total mesorectal excision compared to laparoscopic TME for mid and low rectal cancer—current evidence. Ann Laparosc Endosc Surg 2018;3:41. [Crossref]
- Deijen CL, Tsai A, Koedam TW, et al. Clinical outcomes and case volume effect of transanal total mesorectal excision for rectal cancer: a systematic review. Tech Coloproctol 2016;20:811-24. [Crossref] [PubMed]
- Ito M, Sugito M, Kobayashi A, et al. Relationship between multiple numbers of stapler firings during rectal division and anastomotic leakage after laparoscopic rectal resection. Int J Colorectal Dis 2008;23:703-7. [Crossref] [PubMed]
- Lee L, de Lacy B, Gomez Ruiz M, et al. A Multicenter Matched Comparison of Transanal and Robotic Total Mesorectal Excision for Mid and Low-rectal Adenocarcinoma. Ann Surg 2019;270:1110-6. [Crossref] [PubMed]
- 2017 European Society of Coloproctology (ESCP) collaborating group. An international multicentre prospective audit of elective rectal cancer surgery; operative approach versus outcome, including transanal total mesorectal excision (TaTME). Colorectal Dis 2018;20 Suppl 6:33-46. [Crossref] [PubMed]
- Koedam TW, van Ramshorst GH, Deijen CL, et al. Transanal total mesorectal excision (TaTME) for rectal cancer: effects on patient-reported quality of life and functional outcome. Tech Coloproctol 2017;21:25-33. [Crossref] [PubMed]
- Veltcamp Helbach M, Koedam TWA, Knol JJ, et al. Quality of life after rectal cancer surgery: differences between laparoscopic and transanal total mesorectal excision. Surg Endosc 2019;33:79-87. [Crossref] [PubMed]
- Lelong B, Meillat H, Zemmour C, et al. Short- and Mid-Term Outcomes after Endoscopic Transanal or Laparoscopic Transabdominal Total Mesorectal Excision for Low Rectal Cancer: A Single Institutional Case-Control Study. J Am Coll Surg 2017;224:917-25. [Crossref] [PubMed]
- Lelong B, de Chaisemartin C, Meillat H, et al. A multicentre randomised controlled trial to evaluate the efficacy, morbidity and functional outcome of endoscopic transanal proctectomy versus laparoscopic proctectomy for low-lying rectal cancer (ETAP-GRECCAR 11 TRIAL): rationale and design. BMC Cancer 2017;17:253. [Crossref] [PubMed]
- Deijen CL, Velthuis S, Tsai A, et al. COLOR III: a multicentre randomised clinical trial comparing transanal TME versus laparoscopic TME for mid and low rectal cancer. Surg Endosc 2016;30:3210-5. [Crossref] [PubMed]


