Exceptional response to FOLFIRINOX in a patient with pancreatic cancer and a germline RAD51C mutation
Introduction
Current guidelines for germline genetic testing in patients with pancreatic cancer are limited compared to guidelines available for breast, ovarian, and colon cancer patients. However, identification of heritable deleterious mutations can guide treatment decisions for patients and can impact screening recommendations for family members. BRCA1, BRCA2 and PALB2 are genes, which encode proteins that are part of the Fanconi Anemia pathway and are involved in DNA repair. Germline mutations in these genes are known to be associated with increased risk for pancreatic adenocarcinoma in addition to other types of cancers. Proteins in the Fanconi Anemia pathway function together with a family of proteins known as the RAD51 paralogs which consist of five proteins (RAD51B, RAD51C, RAD51D, XRCC2 and XRCC3). The PALB2 protein acts as a hub to join RAD51 with BRCA1 and BRCA2 to repair double stranded DNA breaks via homologous recombination (1).
We report a patient with a germline RAD51C mutation with pancreatic adenocarcinoma. We pay special attention to his response to systemic therapy with the cross-linking agent oxaliplatin, which supports a causal relationship between the RAD51C mutation and the pathogenesis of his cancer.
Case presentation
A 60-year-old Hispanic man with a past medical history of hypertension, dyslipidemia and gout presented with new onset dyspepsia, and given persistence of his symptoms and an associated 14-pound weight loss. His family history was positive for prostate cancer in his father in his 80s, a paternal cousin with ovarian cancer in her 60s, and a maternal cousin with endometrial cancer. He was referred for an ultrasound of the abdomen. The ultrasound showed a cystic mass in the tail of the pancreas, as well as multiple liver masses and retroperitoneal adenopathy. A computed tomography (CT) scan revealed a hypo-enhancing mass in the pancreatic distal body and tail measuring 6.2×4.6 cm2, encasing the splenic artery and celiac trunk, extensive abdominal lymphadenopathy, numerous scattered pulmonary nodules and numerous hypo-enhancing liver tumors consistent with metastatic pancreatic cancer. He underwent percutaneous biopsy of a liver tumor, which was positive for metastatic poorly differentiated adenocarcinoma, compatible with pancreatobiliary primary.
Next generation sequencing of the tumor biopsy using a commercially available panel revealed alterations in KRAS, ASXL1, EP300 loss, KMT2C (MLL3), and TP53. The tumor was microsatellite stable and the tumor mutation burden was intermediate (8 Muts/Mb). Somatic variants of uncertain significance are shown in Table 1. Multigene panel testing of germline DNA revealed that he carries a mutation in the RAD51C gene (c.709C>T, p.Arg237Ter). Additionally, two variants of uncertain significance were identified; BRCA2 (c.3262C>T, p.Pro1088Ser) and MUTYH (c.1234C>T, p.Arg412Cys), both of which have been reported by other labs as being benign or likely benign variants and therefore are not suspected to be associated with an increased risk for cancer.
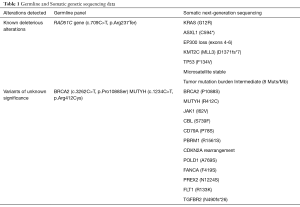
Full table
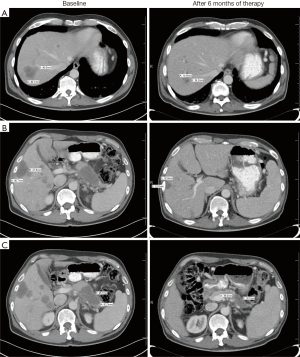
Given his excellent performance status he was started on systemic palliative chemotherapy with FOLFIRINOX. Repeat CT imaging after 6 and 12 cycles showed progressive radiological improvement, with near complete resolution of the pulmonary nodules with one remaining residual measurable subpleural nodule in the left lower lobe measuring 0.6 cm (previously measuring 1 cm), as well as interval decrease in the size and number of his liver lesions (see Figure 1). His pancreatic mass also decreased in size. A PET/CT scan demonstrated near-complete resolution of his disease with remaining increased metabolic activity in the tail of the pancreas with an SUV 2.5–2.9 and in the liver with an SUV 3.7. He remains on chemotherapy and is currently being screened for a clinical trial of a PARP inhibitor as a maintenance strategy. His daughter inherited the RAD51C mutation and has been counseled on prophylactic salpingo-oophorectomy at the age of 45–50 years.
Discussion
The Fanconi Anemia and homologous recombination pathway are activated by replicative stress, particularly DNA damage caused by cross-linking agents or reactive oxygen species. Therefore, a germline mutation in any of the genes in this pathway leads to less effective DNA repair, as shown by its response to DNA damaging agents. Indeed, BRCA1/2 mutation carriers with ovarian or pancreatic cancer show higher response rates to treatment with DNA damaging agents resulting in improved outcome (2,3).
Germline mutations in the RAD51 paralogs have been associated with susceptibility to cancer. Specifically, RAD51C mutations are associated with an estimated increased risk for ovarian cancer corresponding to 9% at age 80 years, with the mean age being approximately 60 years (4). Current NCCN guidelines recommend that prophylactic salpingo-oophorectomy be considered beginning at 45 to 50 years (5). At this time, there is unknown or insufficient evidence for increased risk of other cancer types, though some reports of its contribution to breast cancer in some families exist (3,6).
To our knowledge, this is the first reported case of a RAD51C germline mutation in a patient with pancreatic adenocarcinoma. There are reports of RAD51C mutations in patients with GIST and head and neck cancers (7,8). The patient’s remarkable response to platinum-based therapy supports the hypothesis that the RAD51C mutation contributed to the development of his pancreatic cancer (9,10). Platinum agents exert their antineoplastic activity by causing DNA crosslinking. DNA repair of these crosslinks is dependent on both Fanconi anemia (FA) and BRCA proteins, which act in homologous recombination repair pathways (1,11). Therefore, germline and somatic homologous recombination mutations may be predictive of platinum sensitivity (12). Therefore, the inability of the tumor cells to repair the damage cause by the oxaliplatin could explain his excellent response to FOLFIRINOX.
Conclusions
The case described suggests that carriers of RAD51C mutations may be at increased risk to develop pancreatic cancer and that platinum agents may be a useful therapeutic modality in patients with tumors associated with a germline RAD51C mutation. Germline genetic testing in patients with pancreatic adenocarcinoma might help select better agents for therapy and might also impact screening recommendation for family members.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images. A copy of the written consent is available for review.
References
- Michl J, Zimmer J, Tarsounas M. Interplay between Fanconi anemia and homologous recombination pathways in genome integrity. EMBO J 2016;35:909-23. [Crossref] [PubMed]
- Cass I, Baldwin RL, Varkey T, et al. Improved survival in women with BRCA-associated ovarian carcinoma. Cancer 2003;97:2187-95. [Crossref] [PubMed]
- Dann RB, DeLoia JA, Timms KM, et al. BRCA1/2 mutations and expression: response to platinum chemotherapy in patients with advanced stage epithelial ovarian cancer. Gynecol Oncol 2012;125:677-82. [Crossref] [PubMed]
- Coulet F, Fajac A, Colas C, et al. Germline RAD51C mutations in ovarian cancer susceptibility. Clin Genet 2013;83:332-6. [Crossref] [PubMed]
- Daly MB. National Comprehensive Cancer Network. Genetic/Familial High-Risk Assessment: Breast and Ovarian. Version 1. 10/3/2017; Available online: https://www.nccn.org/professionals/physician_gls/pdf/genetics_screening.pdf
- Jonson L, Ahlborn LB, Steffensen AY, et al. Identification of six pathogenic RAD51C mutations via mutational screening of 1228 Danish individuals with increased risk of hereditary breast and/or ovarian cancer. Breast Cancer Res Treat 2016;155:215-22. [Crossref] [PubMed]
- Scheckenbach K, Baldus SE, Balz V, et al. RAD51C--a new human cancer susceptibility gene for sporadic squamous cell carcinoma of the head and neck (HNSCC). Oral Oncol 2014;50:196-9. [Crossref] [PubMed]
- Shindo K, Yu J, Suenaga M, et al. Deleterious germline mutations in patients with apparently sporadic pancreatic adenocarcinoma. J Clin Oncol 2017;35:3382-90. [Crossref] [PubMed]
- Sidaway P. Bladder cancer: Mutations in DNA-damage-repair pathways confer platinum sensitivity. Nat Rev Urol 2017;14:197. [Crossref] [PubMed]
- Teo MY, O'Reilly EM. Is it time to split strategies to treat homologous recombinant deficiency in pancreas cancer? J Gastrointest Oncol 2016;7:738-49. [Crossref] [PubMed]
- Kim H, D'Andrea AD. Regulation of DNA cross-link repair by the Fanconi anemia/BRCA pathway. Genes Dev 2012;26:1393-408. [Crossref] [PubMed]
- Pennington KP, Walsh T, Harrell MI, et al. Germline and somatic mutations in homologous recombination genes predict platinum response and survival in ovarian, fallopian tube, and peritoneal carcinomas. Clin Cancer Res 2014;20:764-75. [Crossref] [PubMed]


