The nab-paclitaxel/gemcitabine regimen for patients with refractory advanced pancreatic adenocarcinoma
Introduction
Pancreatic cancer (PC) is now the third leading cause of cancer death in the United States with an estimated incidence of 43,920 and 37,390 deaths and is projected to become the second leading cause of cancer death by 2020 (1). Gemcitabine had been the mainstay of chemotherapy for PC for over a decade, as it has demonstrated to be more effective than 5-FU with improved clinical benefit and median survival (2). Erlotinib also showed a statistically significant OS advantage in combination with gemcitabine compared to gemcitabine alone, but the clinical benefit was small and this combination has not been widely accepted (3). More recently two phase III randomized trials have changed practice. The first was the ACCORD/PRODIGE trial that studied the combination of 5-FU, folinic acid (FA), irinotecan and oxaliplatin (FOLFIRINOX); this regimen led to an improvement in objective response rate (RR), progression free survival (PFS) and overall survival (OS) compared to gemcitabine alone (median OS of 11 vs. 6.8 months, P<0.001) (4). The second was the MPACT trial, which investigated the nab-paclitaxel/gemcitabine (AG) combination and also showed an improved RR, OS and PFS compared to gemcitabine alone (median OS of 8.5 vs. 6.7 months respectively, P<0.001) (5). Guidelines now advocate these two regimens as the preferred choices.
Most patients will eventually develop disease recurrence and therefore there is a clear need for effective second line therapies. Multiple agents have been tested for second-line therapy including erlotinib and other targeted therapies, 5-FU, capecitabine, oxaliplatin, and nab-paclitaxel (6-9). The combination of 5-FU and oxaliplatin has shown conflicting results in phase III trials. The CONKO-003 trial demonstrated that oxaliplatin, FA and 5-FU (the OFF regimen) led to a statistically significant improvement in median PFS compared to best supportive care and 5-FU/FA. However, the PANCREOX phase III Trial comparing mFOLFOX vs. infusional 5-FU/FA in gemcitabine-pretreated PC patients showed an improvement in median OS favoring the infusional 5-FU/FA arm over mFOLFOX [9.9 vs. 6.1 months; hazard ratio (HR) =1.78; P=0.02] (10). These results suggested that infusional 5-FU/FA is an alternative for patients with PC previously treated with gemcitabine (8). More recently, the phase III NAPOLI-1 trial demonstrated that the addition of nano-liposomal irinotecan to 5-FU/FA after gemcitabine-based chemotherapy resulted in a significant improvement in median OS vs. 5-FU/FA alone [6.1 vs. 4.2 months; P=0.012; HR =0.67 (95% CI: 0.49–0.92)] (9).
Although the AG regimen is approved for the first-line therapy of PC, there is data supporting the use of this regimen in the second-line setting (7). Herein we present the experience of a single academic tertiary cancer center with the AG combination in progressive metastatic PC, including a significant amount of patients with gemcitabine failure.
Methods
This was a retrospective study consisting of 59 patients with a biopsy-proven metastatic PC, treated with the AG combination as second-line therapy or beyond at the Sylvester Comprehensive Cancer Center in Miami, FL, USA. After IRB-approval was obtained, patients were identified by searching chemotherapy records for patients with a diagnosis of PC who received AG. Patients who received AG as first line therapy were excluded. We stratified the analysis by line of therapy, previous treatment with gemcitabine versus other regimens, and previous adjuvant therapy with gemcitabine. The following information was abstracted from the medical record: demographic data, date of diagnosis, prior treatments, Eastern Cooperative Oncology Group performance status (ECOG PS), CA19-9 levels, radiologic response, adverse events, chemotherapy dose and number of cycles. The toxicities were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events (CTCAE) version 4.0. nab-Paclitaxel 125 mg/m2 and Gemcitabine 1,000 mg/m2 were administered on an outpatient basis, intravenously on days 1, 8 and 15 of a 28-day cycle. Some patients with limited bone marrow reserve after first line therapy were treated with AG every 2 weeks based on anticipated tolerance. Chemotherapy doses were adjusted using the package-insert recommendations, and according to the discretion of the treatment physician when necessary. White blood cell growth factors were also used at the discretion of the treating physician. Treatment was continued until documented progression of disease or development of intolerable toxicity. Responses were assessed according to the Response Evaluation Criteria in Solid Tumors (RECIST) guidelines (11).
The primary endpoint for this analysis was RR. Secondary endpoints included PFS, OS and safety. Patients known to be alive were censored at the time of last contact. All patients who received at least one cycle of AG were included in the analysis. PFS was defined as the duration from the date of the first cycle of AG until the date of documented progression or death, whichever was sooner. OS was defined as the duration from the date of the first cycle of AG until death. PFS and OS were estimated by the Kaplan-Meier method with corresponding two-sided 95% CIs for survival proportions based on Greenwood’s variance and the log-transform method. Statistical significance was defined as P<0.05, and all tests were two-sided. Tests were performed using the IBM SPSS statistics software version 22 (IBM, NY, USA).
Results
Patient characteristics
A total of 59 patients with metastatic PC received AG as second line therapy or beyond between September 2010 and August 2014. Of these, 33 (55%) patients were male and 26 (44%) were female with a median age of 61 years; 4 (7%) patients had an ECOG PS of 0; 36 (61%) had an ECOG PS of 1 and 14 (24%) patients had an ECOG PS of 2; 33 (56%) patients received AG as second line, 14 (24%) as third line, 10 (17%) as fourth line, 1 (1.5%) as fifth line and 1 (1.5%) as sixth line.
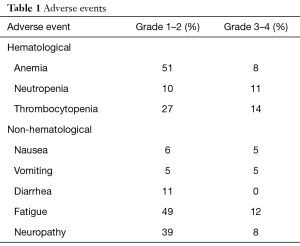
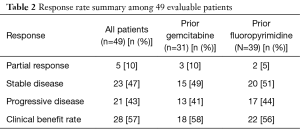
The AG combination was generally well tolerated. The adverse effects were similar to those reported in the MPACT trial and included thrombocytopenia, neutropenia, fatigue and neuropathy as shown in Table 1. At the time of analysis, 28 (57%) patients had clinical benefit with determined as 5 patients (10%) achieving partial responses (PR) and 23 patients (47%) with stable disease (SD) as shown in Table 2. The median OS was 6.5 months (95% CI: 3.9–10.3) and the median PFS was 4.6 months (95% CI: 3–5.1). The OS rate at 6 months was 53% (95% CI: 38–66); the OS rate at 12 months was 27% (95% CI: 14–42).
Full table
Full table
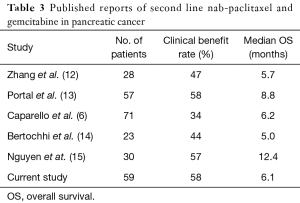
Thirty-four (58%) patients received prior gemcitabine therapy. The most common first line regimen was either the combination of gemcitabine and cisplatin or single agent gemcitabine. The median OS in patients who had received prior treatment with gemcitabine was 6.7 compared to 5.2 months in patients who had not received prior treatment with gemcitabine (95% CI: 4.9–11 months). The median PFS was 4.3 and 4.8 months respectively. The RR comparison of gemcitabine failure patients versus gemcitabine naïve patients is shown in Table 3. Of the 50 patients with measured CA 19-9 levels, 8 (16%) patients had normal levels at the start of treatment 31 (62%) had a decrease in the levels, and 11 (22%) had an increase.
Full table
Discussion
This retrospective analysis suggests that the AG regimen is an active regimen for previously treated PC patients. The combination achieved a clinical benefit rate of 47% in patients who had progressed on first line therapy for metastatic disease. The median PFS and OS were 4.6 and 6.5 months respectively. These results are comparable to the median OS of 6.1 months observed with the combination of nano-liposomal irinotecan plus infusional 5-FU/FA in the Napoli-1 trial (9) and also to the median OS of 4.8 months with the oxaliplatin-based OFF regimen (8). The efficacy observed is especially interesting since 58% of patients had received prior gemcitabine, and even among these, responses were seen, suggesting that nab-paclitaxel can reverse resistance to gemcitabine. The fact that 44% of patients in our study received the AG combination in the third line setting or beyond attests to its feasibility in a difficult-to-treat patient population. This is consistent with the findings of the MPACT trial where patients with a poor performance status still derived benefit from the combination (5).
The toxicities observed in our study were similar to what has been described with first-line AG in the MPACT trial. The most common adverse event was peripheral neuropathy with 45% of patients experiencing any grade neuropathy and 8% with grade 3–4 neuropathy. This high incidence of neuropathy was likely related to the high rate of prior use of cisplatin and oxaliplatin. Most patients required dose modification due to hematologic toxicity when chemotherapy was administered on days 1, 8 and 15 of a 28-day cycle, and some patients were only able to tolerate an every-2-week schedule. In a post-hoc analysis of the MPACT trial, dose reductions and delays in the AG regimen were effective when necessary for toxicity, allowing greater treatment exposure without compromising efficacy.
Although our study is not definitive due to its retrospective nature, lack of control arm and small numbers, our results are consistent with other recent reports showing activity of AG as second-line therapy for metastatic PC as shown in Table 3. Among the published series to date, the clinical benefit rate ranged from 34% to 58% and the median OS ranged from 5 to 12.8 months. Given the emergence of three active combination regimens in the last 5 years (FOLFIRINOX, AG and 5-FU/FA/nano-liposomal irinotecan), the question of sequencing treatments is now being asked for the first time in PC. For those patients who receive AG as first line treatment, the evidence is clear that 5-FU/FA/nano-liposomal irinotecan is a valid second line option. Based on our results and others, we suggest that AG is a reasonable option after first line treatment with FOLFIRINOX. However, further studies are still needed to identify which patients benefit most from the available first and second line options.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: The study was approved by the Institutional Review Board (ID: 00005621).
References
- Siegel R, Naishadham D, Jemal A. Cancer statistics, 2012. CA Cancer J Clin 2012;62:10-29. [Crossref] [PubMed]
- Burris HA 3rd, Moore MJ, Andersen J, et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 1997;15:2403-13. [Crossref] [PubMed]
- Moore MJ, Goldstein D, Hamm J, et al. Erlotinib plus gemcitabine compared with gemcitabine alone in patients with advanced pancreatic cancer: a phase III trial of the National Cancer Institute of Canada Clinical Trials Group. J Clin Oncol 2007;25:1960-6. [Crossref] [PubMed]
- Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 2011;364:1817-25. [Crossref] [PubMed]
- Von Hoff DD, Ervin T, Arena FP, et al. Increased survival in pancreatic cancer with nab-paclitaxel plus gemcitabine. N Engl J Med 2013;369:1691-703. [Crossref] [PubMed]
- Caparello C, Vivaldi C. Second-line therapy for advanced pancreatic cancer: evaluation of prognostic factors and review of current literature. Future Oncol 2016;12:901-8. [Crossref] [PubMed]
- Hosein PJ, De Lima Lopes Jr G, Pastorini VH, et al. A phase ii trial of nab-paclitaxel as second-line therapy in patients with advanced pancreatic cancer. Am J Clin Oncol 2013;36:151-6. [Crossref] [PubMed]
- Oettle H, Riess H, Stieler JM, et al. Second-line oxaliplatin, folinic acid, and fluorouracil versus folinic acid and fluorouracil alone for gemcitabine-refractory pancreatic cancer: Outcomes from the CONKO-003 Trial. J Clin Oncol 2014;32:2423-9. [Crossref] [PubMed]
- Wang-Gillam A, Li CP, Bodoky G, et al. Nanoliposomal irinotecan with fluorouracil and folinic acid in metastatic pancreatic cancer after previous gemcitabine-based therapy (NAPOLI-1): a global, randomised, open-label, phase 3 trial. Lancet 2016;387:545-57. [Crossref] [PubMed]
- Gill S, Ko Y, Cripps C. PANCREOX: A randomized phase 3 study of 5FU/LV with or without oxaliplatin for second-line advanced pancreatic cancer (APC) in patients (pts) who have received gemcitabine (GEM)-based chemotherapy (CT). J Clin Oncol 2014;32:abstr 4022.
- Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009;45:228-47. [Crossref] [PubMed]
- Zhang Y, Hochster H, Stein S, et al. Gemcitabine plus nab-paclitaxel for advanced pancreatic cancer after first-line FOLFIRINOX: single institution retrospective review of efficacy and toxicity. Exp Hematol Oncol 2015;4:29. [Crossref] [PubMed]
- Portal A, Pernot S, Tougeron D, et al. Nab-paclitaxel plus gemcitabine for metastatic pancreatic adenocarcinoma after Folfirinox failure: an AGEO prospective multicentre cohort. Br J Cancer 2015;113:989-95. [Crossref] [PubMed]
- Bertocchi P, Abeni C, Meriggi F, et al. Gemcitabine Plus Nab-Paclitaxel as Second-Line and Beyond Treatment for Metastatic Pancreatic Cancer: a Single Institution Retrospective Analysis. Rev Recent Clin Trials 2015;10:142-5. [Crossref] [PubMed]
- Nguyen KT, Kalyan A, Beasley HS, et al. Gemcitabine/nab-paclitaxel as second-line therapy following FOLFIRINOX in metastatic/advanced pancreatic cancer-retrospective analysis of response. J Gastrointest Oncol 2017;8:556-65. [Crossref] [PubMed]


