Bridging therapy effectiveness in the treatment of hepatocellular carcinoma prior to orthotopic liver transplantation
Introduction
It is estimated that there will be more than 39,000 new cases of liver and intrahepatic bile duct cancer this year in the United States, and more than 25,000 people will die of these diseases (1). An increase in new hepatocellular carcinoma (HCC) cases can be attributed to the aging patient cohort with chronic hepatitis C virus infection (2). HCC is the fastest-growing cause of cancer-related death in United States, with an overall 5-year survival of less than 12% in patients who do not undergo orthotopic liver transplantation (OLT) (2). Worldwide, HCC is the 5th most common cancer and the third most common cause of cancer-related death (3,4).
Currently, OLT is the only curative treatment for patients with cirrhosis, portal hypertension and early-stage HCCs that meet Milan criteria (single lesion ≤5 cm or up to three lesions each ≤3 cm) (5). Since the Milan criteria began to be used to determine eligibility of patients with HCC for OLT, the 5-year survival rate after surgery has increased to 70–80% (6,7). Although OLT may be a successful treatment for patients with cirrhosis and HCC, there are many considerable challenges to transplantation. By far one of the biggest challenges to OLT for patients with HCC is the shortage of donor organs and prolonged waiting times until transplantation. While patients await transplantation, a variety of bridging therapies (BTs) have been used in an effort to maintain disease control including; radiofrequency ablation (RFA), transcatheter arterial chemoembolization (TACE), microwave ablation (MWA), selective internal radiation therapy (SIRT) and stereotactic body radiation therapy (SBRT) varies. The goal of this study was to assess the pathologic response on explant analysis for patients who undergo BT prior to OLT.
Methods
We retrospectively reviewed the clinical data in all 69 patients who underwent OLT for HCC between April, 2009 and December, 2014 at Montefiore Medical Center, Bronx, NY. During this period, 50 patients underwent a total of 125 treatment sessions prior to OLT. Nineteen patients were excluded because they did not receive BTs prior to transplant. The diagnosis of HCC was made using standard imaging criteria; lesions with equivocal imaging findings had biopsy confirmation of their diagnosis.
The study included 37 men and 13 women. The most common etiology of cirrhosis was hepatitis C virus infection, which was present in 44 of the 50 patients (88%). Two patients had cryptogenic cirrhosis, four patients had hepatitis B virus infection and four patients had alcoholic cirrhosis. Twenty-five of the 50 patients had multiple nodules treated with BT. The rest had solitary nodules.
Treatment decisions were made by the members of the institutional Multidisciplinary Tumor Board that included oncologists, hepatologists, radiologists, radiation oncologist and surgeons. BTs included TACE, RFA, MWA, ETOH, SBRT and SIRT. Complete response was defined as the absence of standard radiographic or magnetic resonance (MR) features of HCC following the patient’s last BT. The last imaging available immediately prior to transplant was compared to explant pathology. Tumor necrosis was classified as 0%, <80%, >80% or 100% on the basis of imaging. If necrosis was present but further quantification was not available, nodules were categorized conservatively as <80% necrosis. Tumor response based on imaging was compared to tumor necrosis on explant pathology.
Results
Ninety-three nodules in 50 patients were evaluated after BT. Untreated nodules identified either on imaging or explant pathology were excluded from the analysis. Patients had from 1 to 7 BTs (mean =2.5). Eighty-three nodules received therapy with TACE alone or TACE in combination with another therapy (Table 1).
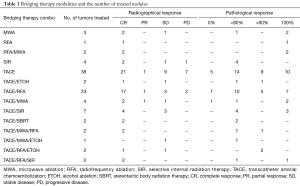
Full table
On radiographic examination, 59 of the 93 tumor nodules had a complete imaging response to therapy. One-third (28 of the 93) of nodules had complete necrosis on pathological examination. Of the nodules with a complete pathological response, 10 had been treated with TACE alone, 7 with TACE and RFA, and 3 with TACE and SIR. The other nodules with a complete pathological response were treated with a variety of other BTs (Table 1).
Only 6 of 50 patients (12%) had complete necrosis of all tumors on pathologic examination after BT. Four of the 6 patients had solitary nodules, and 2 patients each had two nodules. Table 2 gives tumor nodules dimension and BT used for the six patients with complete tumor necrosis (CTN). Of 22 patients with a complete imaging response to BT, 17 (77%) had viable tumor in their explants. One patient had viable tumor on imaging but no viable tumor on pathology.

Full table
Discussion
Many BTs have been used to slow the progression of HCC prior to transplantation or to downstage HCC to within Milan criteria. Of these treatments, TACE and RFA have been the most studied BTs, but newer therapies such as SIRT and SBRT are also being used (8-10). Five-year survival of patients after TACE prior to transplantation ranged from 51% to 93% (11-13). Studies have shown that TACE improves survival in patients with unresectable HCC who are not transplant candidates (14,15). One-year survival after TACE can be up to 85%, with a median overall survival of ranging from 20 to 36 months (16,17). In comparison, patients with inoperable HCC have a 5-year survival of 27% to 61% after RFA (18,19). In one series, however, 44% of patients had satellite nodules after RFA (20). The majority of our patients underwent TACE, RFA or combination TACE and RFA prior to transplant.
Patients treated with other BTs such as MWA, have a survival similar to that of patients treated with TACE and RFA (21). Patient treated with newer therapies such as SBRT and SIRT may have less rapid tumor progression as compared to individuals treated with TACE (22-24). In our study, a total of 15 nodules were treated with BTs that included the newer modalities of SIRT and SBRT. Complete necrosis of four nodules (27%) after SIRT was documented on explant. The remainder of the nodules had <80% necrosis on explant.
Although the literature suggests that BTs are associated with improved tumor-free survival after transplantation, the benefit of BT prior to liver transplantation is unclear when evaluating pathologic response treatment. Published results of examinations of explanted livers following BTs have shown that the extent of tumor necrosis varies widely, ranging from 0–100% (20,25-28). The effectiveness of TACE in treating 27 tumor nodules was assessed by examination of explants that showed 64% mean necrosis; 12 (44%) had 99–100% tumor necrosis (27). More recent studies have documented CTN of approximately 50% of nodules after TACE or TACE and RFA (29,30). In our patients, approximately 30% of nodules had CTN after BT. This data suggests that most patients still have macroscopic HCC after BT, confirming the necessity of OLT as the best chance for cure.
Four of the six patients in our cohort who had CTN initially had solitary nodules. Numerous patients had successfully treated nodules but developed satellite nodules or multinodular disease. Schroeder et al. reported similar findings: 38% of patients had tumor progression on explant that was not seen on imaging (31). Unfortunately, imaging may underestimate residual or recurrent disease after BT (32-34). Our data supports this finding; the majority of our patients who had a complete response to treatment on imaging had viable tumor in their explants. The goal of BT is to achieve adequate tumor control in order for patients to remain eligible for OLT. Given lengthy waitlists for patients awaiting OLT, this strategy is often imperative. These findings have important implications for patients with HCC who are otherwise not OLT candidates; given the high likelihood of residual disease following BT, these patients must be managed aggressively. Current BTs tend not to produce complete responses, and better treatment modalities or sequencing are required in order to improve patient outcomes.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Ethical Statement: Montefiore Medical Center Institutional Review Board approval was obtained for this study (IRB No. 2014-3588). Hospital medical records were used to retrieve patient data and patient’s personal data has been secured.
References
- Howlader N, Noone AM, Krapcho M, et al. editors. SEER Cancer Statistics Review, 1975-2013. National Cancer Institute. Available online: https://seer.cancer.gov/archive/csr/1975_2013/#contents
- Mittal S, El-Serag HB. Epidemiology of hepatocellular carcinoma: consider the population. J Clin Gastroenterol 2013;47 Suppl:S2-6. [Crossref] [PubMed]
- Altekruse SF, McGlynn KA, Reichman ME. Hepatocellular carcinoma incidence, mortality, and survival trends in the United States from 1975 to 2005. J Clin Oncol 2009;27:1485-91. [Crossref] [PubMed]
- Ryerson AB, Eheman CR, Altekruse SF, et al. Annual Report to the Nation on the Status of Cancer, 1975-2012, featuring the increasing incidence of liver cancer. Cancer 2016;122:1312-37. [Crossref] [PubMed]
- Mazzaferro V, Regalia E, Doci R, et al. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis. N Engl J Med 1996;334:693-9. [Crossref] [PubMed]
- Bruix J, Llovet JM. Prognostic prediction and treatment strategy in hepatocellular carcinoma. Hepatology 2002;35:519-24. [Crossref] [PubMed]
- Mazzaferro V, Llovet JM, Miceli R, et al. Predicting survival after liver transplantation in patients with hepatocellular carcinoma beyond the Milan criteria: a retrospective, exploratory analysis. Lancet Oncol 2009;10:35-43. [Crossref] [PubMed]
- Dekervel J, van Malenstein H, Vandecaveye V, et al. Transcatheter arterial chemoembolization with doxorubicin-eluting superabsorbent polymer microspheres in the treatment of hepatocellular carcinoma: midterm follow-up. J Vasc Interv Radiol 2014;25:248-55.e1. [Crossref] [PubMed]
- Hołówko W, Wróblewski T, Wojtaszek M, et al. Transarterial Chemoembolization Prior to Liver Transplantation in Patients with Hepatocellular Carcinoma. Ann Transplant 2015;20:764-8. [Crossref] [PubMed]
- Golfieri R, Cappelli A, Cucchetti A, et al. Efficacy of selective transarterial chemoembolization in inducing tumor necrosis in small (<5 cm) hepatocellular carcinomas. Hepatology 2011;53:1580-9. [Crossref] [PubMed]
- Graziadei IW, Sandmueller H, Waldenberger P, et al. Chemoembolization followed by liver transplantation for hepatocellular carcinoma impedes tumor progression while on the waiting list and leads to excellent outcome. Liver Transpl 2003;9:557-63. [Crossref] [PubMed]
- Decaens T, Roudot-Thoraval F, Bresson-Hadni S, et al. Impact of pretransplantation transarterial chemoembolization on survival and recurrence after liver transplantation for hepatocellular carcinoma. Liver Transpl 2005;11:767-75. [Crossref] [PubMed]
- Zhang Z, Liu Q, He J, et al. The effect of preoperative transcatheter hepatic arterial chemoembolization on disease-free survival after hepatectomy for hepatocellular carcinoma. Cancer 2000;89:2606-12. [Crossref] [PubMed]
- Lo CM, Ngan H, Tso WK, et al. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology 2002;35:1164-71. [Crossref] [PubMed]
- Llovet JM, Real MI, Montaña X, et al. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet 2002;359:1734-9. [Crossref] [PubMed]
- Terzi E, Piscaglia F, Forlani L, et al. TACE performed in patients with a single nodule of hepatocellular carcinoma. BMC Cancer 2014;14:601. [Crossref] [PubMed]
- Brown KT, Do RK, Gonen M, et al. Randomized Trial of Hepatic Artery Embolization for Hepatocellular Carcinoma Using Doxorubicin-Eluting Microspheres Compared With Embolization With Microspheres Alone. J Clin Oncol 2016;34:2046-53. [Crossref] [PubMed]
- Lee DH, Lee JM, Lee JY, et al. Radiofrequency ablation of hepatocellular carcinoma as first-line treatment: long-term results and prognostic factors in 162 patients with cirrhosis. Radiology 2014;270:900-9. [Crossref] [PubMed]
- N'Kontchou G, Mahamoudi A, Aout M, et al. Radiofrequency ablation of hepatocellular carcinoma: long-term results and prognostic factors in 235 Western patients with cirrhosis. Hepatology 2009;50:1475-83. [Crossref] [PubMed]
- Brillet PY, Paradis V, Brancatelli G, et al. Percutaneous radiofrequency ablation for hepatocellular carcinoma before liver transplantation: a prospective study with histopathologic comparison. AJR Am J Roentgenol 2006;186:S296-305. [Crossref] [PubMed]
- Huo YR, Eslick GD. Microwave Ablation Compared to Radiofrequency Ablation for Hepatic Lesions: A Meta-Analysis. J Vasc Interv Radiol 2015;26:1139-46.e2. [Crossref] [PubMed]
- Bibault JE, Dewas S, Vautravers-Dewas C, et al. Stereotactic body radiation therapy for hepatocellular carcinoma: prognostic factors of local control, overall survival, and toxicity. PLoS One 2013;8:e77472. [Crossref] [PubMed]
- Salem R, Gordon AC, Mouli S, et al. Y90 Radioembolization Significantly Prolongs Time to Progression Compared With Chemoembolization in Patients With Hepatocellular Carcinoma. Gastroenterology 2016;151:1155-63.e2. [Crossref] [PubMed]
- Nugent FW, Qamar A, Stuart KE, et al. A randomized phase II study of individualized stereotactic body radiation therapy (SBRT) versus transarterial chemoembolization (TACE) with DEBDOX beads as a bridge to transplant in hepatocellular carcinoma (HCC). J Clin Oncol 2015;35:abstr 223.
- Mazzaferro V, Battiston C, Perrone S, et al. Radiofrequency ablation of small hepatocellular carcinoma in cirrhotic patients awaiting liver transplantation: a prospective study. Ann Surg 2004;240:900-9. [Crossref] [PubMed]
- Fontana RJ, Hamidullah H, Nghiem H, et al. Percutaneous radiofrequency thermal ablation of hepatocellular carcinoma: a safe and effective bridge to liver transplantation. Liver Transpl 2002;8:1165-74. [Crossref] [PubMed]
- Wong LL, Tanaka K, Lau L, et al. Pre-transplant treatment of hepatocellular carcinoma: assessment of tumor necrosis in explanted livers. Clin Transplant 2004;18:227-34. [Crossref] [PubMed]
- Maddala YK, Stadheim L, Andrews JC, et al. Drop-out rates of patients with hepatocellular cancer listed for liver transplantation: outcome with chemoembolization. Liver Transpl 2004;10:449-55. [Crossref] [PubMed]
- Chedid MF, Scaffaro LA, Chedid AD, et al. Transarterial Embolization and Percutaneous Ethanol Injection as an Effective Bridge Therapy before Liver Transplantation for Hepatitis C-Related Hepatocellular Carcinoma. Gastroenterol Res Pract 2016;2016:9420274. [PubMed]
- Frenette CT, Osorio RC, Stark J, et al. Conventional TACE and drug-eluting bead TACE as locoregional therapy before orthotopic liver transplantation: comparison of explant pathologic response. Transplantation 2014;98:781-7. [Crossref] [PubMed]
- Schroeder T, Sotiropoulos GC, Molmenti EP, et al. Changes in staging for hepatocellular carcinoma after radiofrequency ablation prior to liver transplantation as found in the explanted liver. Hepatogastroenterology 2011;58:2029-31. [PubMed]
- Pompili M, Pacella CM, Francica G, et al. Percutaneous laser ablation of hepatocellular carcinoma in patients with liver cirrhosis awaiting liver transplantation. Eur J Radiol 2010;74:e6-11. [Crossref] [PubMed]
- Yao FY, Kerlan RK Jr, Hirose R, et al. Excellent outcome following down-staging of hepatocellular carcinoma prior to liver transplantation: an intention-to-treat analysis. Hepatology 2008;48:819-27. [Crossref] [PubMed]
- Marin HL, Furth EE, Olthoff K, et al. Histopathologic outcome of neoadjuvant image-guided therapy of hepatocellular carcinoma. J Gastrointestin Liver Dis 2009;18:169-76. [PubMed]


