Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy combined with two-stage hepatectomy for multiple and bilobar desmoplastic small round cell tumor liver metastases
Introduction
Desmoplastic small round cell tumor (DSRCT) is a rare, highly aggressive malignant mesenchymal tumor initially described in 1981 by Gerald and Rosai (1). DSRCT is most commonly seen in young patients and rarely in adults. It has a predilection for the serosal surface, most often the abdominal and pelvic cavity. At the time of presentation, up to 50% of the patients may have extra-peritoneal metastasis and even higher rates of local metastasis (2-5). The most common presenting symptom is abdominal pain, followed by abdominal distention, abdominal mass, and hepatomegaly (4,5).
There is a lack of consensus regarding the management of DSRCT. Given the rareness of the disease, there are no large randomized controlled trials in the management of this tumor. There is also no reported data on the management of liver metastasis from this disease. Multimodal management including cytoreductive surgery (CRS), chemotherapy, and radiotherapy has been shown to be effective in the treatment of DSRCT (2,3). Also, recent studies have reported the benefit from hyperthermic intraperitoneal chemotherapy (HIPEC) in this disease for intraperitoneal metastatic disease (6).
The liver is the most common site of extra-peritoneal metastasis. To our knowledge, there is no published data regarding surgical management of metastatic bi-lobar hepatic lesions from DSRCT. In this report, we present a case of an abdominal DSRCT in an adult patient with peritoneal and hepatic metastatic disease. The patient received a multimodal treatment, including CRS, HIPEC and two-stage hepatectomy (TSHP).
Case presentation
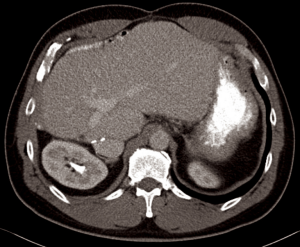
A 51-year-old male presented with a history of new onset reflux and intermitted mild abdominal pain of nondescript origin in October 2014. His liver function tests and tumor markers including alpha-fetoprotein and carcinoembryonic antigen were within normal limit. A computer tomography (CT) scan showed a large tumor in the left upper abdomen with multiple bi-lobar liver metastases (Figure 1). A biopsy of the abdominal mass was consistent with a DSRCT of the peritoneal cavity. Given these findings, the patient was treated with multi-modal therapy including CRS, HIPEC, TSHP combined with (90Y) Yittrium-90 radioembolization of the right hepatic lobe and systemic chemotherapy.
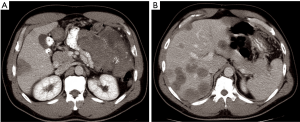
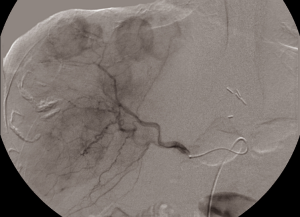
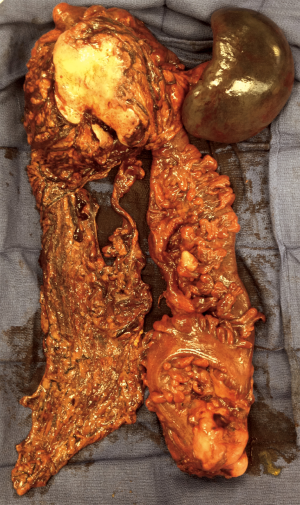
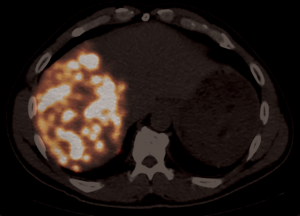
After informed consent, the patient was taken to the operating room in November 2014. A large mass was appreciated in the left upper quadrant over the splenic flexure of the colon extending towards the tail of the pancreas and multiple lesions along the colon extending towards the pelvis. There were multiple lesions in the right lobe of the liver with two lesions noted in the left lateral segment corresponding to the CT images. CRS with tumor debulking and excision of peritoneal metastases and mesenteric metastases was performed. This included an en bloc resection of the tumor to include the entire colon, the distal pancreas, the spleen and the omentum (Figure 2). There was no residual macroscopic disease at the end of the surgery—CC 0 (Sugarbaker classification) (2). A partial liver resection of two liver metastases in segment II and III was performed as part of the first stage of TSHP with a goal to clear the left hemi-liver of all metastases in the first stage. Concomitantly, HIPEC with 100 mg of cisplatin was performed for 60 minutes. Histopathology from both en-block colonic and liver tumor confirmed it as DSRCT. The distal margin of the colon was clear of tumor. 18F-FDG PET-CT in December 2014 demonstrated metabolic response corresponding to the area associated with metastases in the right lobe (Figure 3). (90Y) Yttrium radioembolization was done through the right hepatic artery for right hepatic lobe metastases in December 2016 (Figure 4). Patient’s left lobe liver volume was 766 cc (increased from 33% to 38% of the total liver volume) 6 weeks after radioembolization (Figure 5). The patient underwent extended right hepatectomy including segment IV and cholecystectomy in February 2015. There was no portal clamping during the procedure. The histopathology confirmed it as metastatic DSRCT.
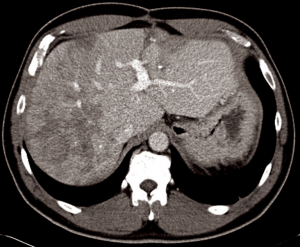
The patient had a right-sided pleural empyema one month after the surgery for which he required right-sided video-assisted thoracoscopy and decortication in March 2015. The patient recovered well after this episode. He was given four cycles of AIM (Doxorubicin & Ifosfamide) chemotherapy between September and November 2015. A follow-up CT scan at 6, 12, 18 and 24 months after his final surgery showed no tumor recurrence (Figure 6). From 2nd year the patient will be followed up annually with CT scans for 5 years.
Discussion
DSRCT is an aggressive tumor of mesenchymal origin mostly found in the pediatric population and young adults. Patients are typically from 5–30 years, and 90% are men (7). The most common presenting symptoms are abdominal pain and abdominal mass. Occasionally, a patient presents with constipation, ascites or urinary symptoms. At the time of diagnosis, the disease is almost always locally disseminated, and 50% of the patients have an extraperitoneal metastatic disease. The liver is the most common site of extra-peritoneal disease, followed by lung and spleen (2,3).
The overall prognosis of this disease is poor, with a mean overall survival of 15% to 25% (3-8). Surgical resection, adjuvant chemotherapy, and radiotherapy may increase the overall survival, by 30–40 months. The combination of these therapies has been shown to have better outcomes than individual therapy by itself (2-9). DSRCT is a member of the large family of small round cell tumors of childhood, together with PNET (Ewing sarcoma), alveolar and embryonal rhabdomyosarcoma, poorly differentiated synovial sarcoma and rhabdoid tumors (10). DSRCT is now identified as a polyphenotypic lesion with epithelial, muscular, and neuronal biomarkers (11). Its polyphenotypic nature led some authors to consider DSRCT almost as carcinomas, and thus, CRS was performed. CRS has been shown to improve survival (12). Patients with a lower peritoneal carcinomatosis index (PCI) and complete CRS (resection of >90% of the disease), have a significantly higher overall survival rate than those with incomplete resection of the tumor and higher PCI (7-12). HIPEC has been shown to benefit patients with disseminated peritoneal malignancies such as colorectal cancer, appendiceal cancer, and mesothelioma, with acceptable complication rates. Although some studies have shown no significant difference in overall survival with HIPEC in DSCRT, others have shown benefit in the treatment of peritoneal sarcomatosis, especially DSCRT (6,12,13). Even though it is an aggressive tumor it has been demonstrated to be chemosensitive (4). Subbiah et al. reported the case of a chemo-resistant DSRCT with hepatic metastasis successfully treated with (90Y) Yttrium microspheres with an evident metabolic response on PET-CT (14). (90Y) Yttrium microspheres were used in our patient for radioembolization of the right lobe with the view of not only increasing the future liver remnant but also treating the right lobe metastases in case the patient becomes an unresectable candidate.
Recurrence rates (RR) in DSRCT are high. Some authors report RR from 50% to 100% (2,4,5,7,9), with mean times to recurrence from 12 to 26 months. Honoré et al. reported 1 and 3-year disease free survival (DFS) rates of 41.6% and 16.7% respectively, with a median DFS of 15 months. Also, patients with disease outside of the abdominal cavity had worse DFS but did not differ in terms of overall survival (6). Our patient remains disease free at 2 years follow-up.
As previously described, the liver is the most common site of metastatic disease from DSCRT, and there is a high incidence of metastatic disease at the time of diagnosis. Even though there are reports from liver resections of metastatic lesions from this tumor, there is a paucity of data reported on its management and outcomes. In a series reported by Honoré et al., the three patients who had bi-lobar liver metastases but had complete CRS, had a median survival of 14.8 months. They concluded surgery to be inappropriate for this group of patients and recommended chemotherapy (2). TSHP with portal vein embolization has been successful in treating bi-lobar colorectal liver metastases when there is a concern for a small future liver remnant. Jaeck et al. quoted the 1 and 3-year overall survival rates as 70.0% and 54.4% in the 25 patients with whom they completed TSHP combined with portal vein embolization for bilobar colorectal liver metastases (BCLM) (15). More recently, Karoui et al. quoted the 3 and 5-year overall survival rates as 80% and 48% in patients undergoing TSHP for BCLM (16). It is a safe and effective procedure with similar outcomes as single-stage resection. However, no data has been published for DSRCT (15,17).
Our patient had a presentation and behavior similar to most published data on DSCRT, but it was unique in several ways. The patient was above the average age of the patient population with this rare disease. The patient had an advanced stage at the time of diagnosis, with tumor involving the abdominal cavity and both lobes of the liver. Multi-modal therapy with complete CRS, HIPEC, TSHP combined with (90Y) Yittrium-90 radioembolization of the right hepatic lobe, and systemic chemotherapy was successful even in the background of bi-lobar liver metastases. To our knowledge, this is the first report of a TSHP for a patient with liver metastasis for DSRCT. The patient remains alive and disease free at 24 months since his final surgery.
Given the rareness of this disease, there is a lack of data and consensus regarding the management of this disease, with no available large randomized controlled trials. Even though we present a successful report of multimodal management including TSHP for DSCRT, there is a need for more investigation to prove the outcomes and benefits of this procedure. In the meantime, we will have to rely on retrospective and case studies for this rare disease.
Conclusions
Multimodal management of DSRCT seems to be the best option for patients with this disease. The presence of bi-lobar liver metastasis shouldn’t categorize patients as unresectable, and preclude them from curative options. TSHP seems to be a feasible and safe option in selected cases, with a potentially good outcome.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this Case report and any accompanying images.
References
- Gerald WL, Miller HK, Battifora H, et al. Intra-abdominal desmoplastic small round-cell tumor. Report of 19 cases of a distinctive type of high-grade polyphenotypic malignancy affecting young individuals. Am J Surg Pathol 1991;15:499-513. [Crossref] [PubMed]
- Honoré C, Amroun K, Vilcot L, et al. Abdominal desmoplastic small round cell tumor: multimodal treatment combining chemotherapy, surgery, and radiotherapy is the best option. Ann Surg Oncol 2015;22:1073-9. [Crossref] [PubMed]
- Lal DR, Su WT, Wolden SL, et al. Results of multimodal treatment for desmoplastic small round cell tumors. J Pediatr Surg 2005;40:251-5. [Crossref] [PubMed]
- Bisogno G, Roganovich J, Sotti G, et al. Desmoplastic small round cell tumour in children and adolescents. Med Pediatr Oncol 2000;34:338-42. [Crossref] [PubMed]
- Hassan I, Shyyan R, Donohue JH, et al. Intraabdominal desmoplastic small round cell tumors: a diagnostic and therapeutic challenge. Cancer 2005;104:1264-70. [Crossref] [PubMed]
- Hayes-Jordan A, Green H, Lin H, et al. Cytoreductive surgery and Hyperthermic Intraperitoneal Chemotherapy (HIPEC) for children, adolescents, and young adults: the first 50 cases. Ann Surg Oncol 2015;22:1726-32. [Crossref] [PubMed]
- Hayes-Jordan A, Anderson PM. The diagnosis and management of desmoplastic small round cell tumor: a review. Curr Opin Oncol 2011;23:385-9. [Crossref] [PubMed]
- Gerald WL, Ladanyi M, de Alava E, et al. Clinical, pathologic, and molecular spectrum of tumors associated with t(11;22)(p13;q12): desmoplastic small round-cell tumor and its variants. J Clin Oncol 1998;16:3028-36. [Crossref] [PubMed]
- Zhang J, Xu H, Ren F, et al. Analysis of clinicopathological features and prognostic factors of desmoplastic small round cell tumor. Pathol Oncol Res 2014;20:161-8. [Crossref] [PubMed]
- Dufresne A, Cassier P, Couraud L, et al. Desmoplastic small round cell tumor: current management and recent findings. Sarcoma 2012;2012:714986. [Crossref] [PubMed]
- Thway K, Noujaim J, Zaidi S, et al. Desmoplastic Small Round Cell Tumor: Pathology, Genetics, and Potential Therapeutic Strategies. Int J Surg Pathol 2016;24:672-84. [Crossref] [PubMed]
- Hayes-Jordan A, Green HL, Lin H, et al. Complete cytoreduction and HIPEC improves survival in desmoplastic small round cell tumor. Ann Surg Oncol 2014;21:220-4. [Crossref] [PubMed]
- Msika S, Gruden E, Sarnacki S, et al. Cytoreductive surgery associated to hyperthermic intraperitoneal chemoperfusion for desmoplastic round small cell tumor with peritoneal carcinomatosis in young patients. J Pediatr Surg 2010;45:1617-21. [Crossref] [PubMed]
- Subbiah V, Murthy R, Anderson PM. [90Y]yttrium microspheres radioembolotherapy in desmoplastic small round cell tumor hepatic metastases. J Clin Oncol 2011;29:e292-4. [Crossref] [PubMed]
- Jaeck D, Oussoultzoglou E, Rosso E, et al. A two-stage hepatectomy procedure combined with portal vein embolization to achieve curative resection for initially unresectable multiple and bilobar colorectal liver metastases. Ann Surg 2004;240:1037-49; discussion 1049-51. [Crossref] [PubMed]
- Karoui M, Vigano L, Goyer P, et al. Combined first-stage hepatectomy and colorectal resection in a two-stage hepatectomy strategy for bilobar synchronous liver metastases. Br J Surg 2010;97:1354-62. [Crossref] [PubMed]
- Huang SY, Aloia TA, Shindoh J, et al. Efficacy and safety of portal vein embolization for two-stage hepatectomy in patients with colorectal liver metastasis. J Vasc Interv Radiol 2014;25:608-17. [Crossref] [PubMed]