Utilization and trends in palliative therapy for stage IV pancreatic adenocarcinoma patients: a U.S. population-based study
Introduction
With approximately 50% of cases presenting with metastatic disease, pancreatic adenocarcinoma continues to carry a dismal prognosis (1,2). Its gravity is twofold: the lack of effective screening protocols leads many patients to be diagnosed with unresectable disease, and current limitations with surgical and medical therapies typically preclude effective treatment once diagnosed. Unlike diseases such as metastatic colorectal cancer, patients with metastatic pancreatic cancer are currently not candidates for curative resection. Until the effectiveness of available therapies can surpass the aggressive biology of the disease, palliation of symptoms will remain a mainstay of treatment, with a focus on alleviating symptoms rather than treating the disease. While not all patients present with symptoms requiring intervention, they frequently develop with disease progression (2,3).
Because of the location of the pancreas within the retroperitoneum, its proximity to vital structures, aggressive behavior, and potential for metastatic spread, pancreatic adenocarcinoma can cause a variety of symptoms that can impair a patient’s quality of life. Pain is a presenting symptom in approximately half of patients, and develops in a majority of patients at some point during the disease process (2,4,5). Furthermore, it has been estimated that approximately 80% of patients with pancreatic head adenocarcinoma will seek intervention for symptoms of biliary obstruction (2). Gastric outlet obstruction, often requiring intervention, occurs in 20% of patients, and may develop in another 20–40% during the course of their disease (2). The challenge with palliation is minimizing symptoms of disease while limiting morbidity due to therapy. Benefits of palliation extend beyond alleviating pain; by shifting the clinical focus to palliative strategies, several studies have noted lower overall costs of care (6,7). Understanding the current status of palliative care among patients with pancreatic cancer is important for addressing existing barriers to its implementation. This study focused on the cohort of patients who presented with metastatic pancreatic adenocarcinoma at diagnosis, and aimed to evaluate trends and patterns of palliative care utilization in the United States.
Methods
Data
The study cohort was derived from the National Cancer Data Base (NCDB) Participant User File. The NCDB is a national database that captures approximately 70% of incident cancer cases in the United States. All cases of pancreatic adenocarcinoma that presented with clinical stage IV disease from 2003 to 2011 were abstracted from the database. Interventions that were aimed at palliating or alleviating symptoms were denoted “palliative”. Patients were stratified into those who underwent a specified type of palliative therapy (surgery, radiation, systemic chemotherapy, pain management, or a combination thereof), those who received a referral for palliative therapy, or those without intervention or referral.
Covariates
Covariates of interest included patient-specific factors such as age (stratified into age groups 18–59, 60–69, 70–79, or ≥80 years), sex, race/ethnicity (white non-Hispanic, black non-Hispanic, Hispanic, or other), insurance status (private, Medicare, Medicaid, uninsured, or unknown), quartile of median household income, and number of Charlson/Deyo comorbidities (0, 1, or ≥2). Hospital-specific covariates included facility type (community cancer program, comprehensive community cancer program, academic/research program, or other) and geographic region of the United States (Northeast, South, Midwest, and West).
Statistical analysis
Demographic and facility-related covariates were compared between the group that received a specified palliative intervention and the group that did not using standard univariate statistics. Regression analyses were used to identify significant trends in use of palliative therapy over the time period of the cohort. Multivariable logistic regression was used to model receipt of palliative therapy as a function of the aforementioned covariates. Kaplan-Meier curves were constructed for the groups undergoing various palliative interventions. In order to determine the association of the various palliative- and non-palliative interventions with patient survival, multivariable survival analyses were performed using a Weibull model because survival was not consistent with a proportional hazards assumption. All analyses were performed using STATA software (version 12.1, StataCorp, College Station, TX, USA). Statistical significance was defined as P<0.05.
Results
Study cohort
Within the primary study cohort, a total of 68,075 patients diagnosed with stage IV pancreatic adenocarcinoma were identified; of these, 11,449 (16.8%) received designated palliative therapy. Among this subset, the largest proportion received systemic chemotherapy (37.2%), followed by surgery (19.0%), pain management alone (15.3%), radiation (8.1%), or a combination thereof (8.7%). Compared to the 16.8% of patients receiving designated palliative therapy, rates of utilization of non-palliative therapy were as follows: systemic chemotherapy (43.6%), surgery (0.6%), radiation (1.4%), and combination therapy (4.6%). Approximately 11.7% had a referral for palliative care, but no specific interventions were identified. Univariate comparisons between those receiving and not receiving designated palliative therapy are shown in Table 1.
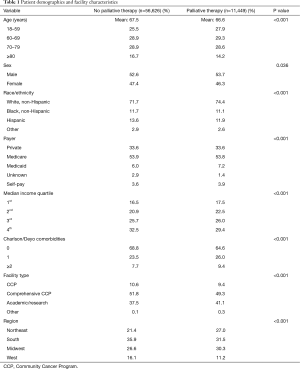
Full table
Trends
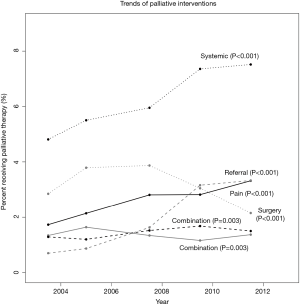
Among patients with stage IV disease, there was a significant increase in the utilization of palliative care, from 12.9% in 2003 to 19.2% in 2011 (P<0.001). Considering the various interventions individually, the rate of surgery significantly decreased over this time interval, while the rate of radiation therapy utilization remained stable (Figure 1). Systemic therapy, pain management strategies, and combination therapy all significantly increased over this time interval (Figure 1). Referral for palliative care also increased, although the type of therapy that was performed, if at all, was unable to be determined among this subgroup.
Logistic regression
After controlling for baseline differences between the cohorts, multivariable logistic regression showed that increasing age was associated with lower odds of receiving palliative therapy, particularly among those over 80 years (Table 2). The difference in utilization between males and females was not statistically significant (P=0.335). Being of black race or Hispanic ethnicity, however, was associated with lower utilization of palliative measures [odds ratio (OR) 0.83 with 95% confidence interval (CI) 0.77 to 0.89, and OR 0.80 with 95% CI: 0.75 to 0.85, respectively] versus Caucasians. Presence of associated comorbidities increased the likelihood of receiving palliative therapy, with 16% higher odds in those with one comorbidity (95% CI: 1.10 to 1.21), and 27% higher odds in those with two or more comorbidities (95% CI: 1.18 to 1.37). Compared to patients with government insurance, utilization was lower for privately insured patients (OR 0.90; 95% CI: 0.85 to 0.96). Comprehensive community and academic centers were more likely to offer palliative therapies than community cancer centers (Table 2). Additionally, significant regional variation was noted, with the Northeast region of the country having the highest utilization compared to the South, Midwest, or West (Table 2).
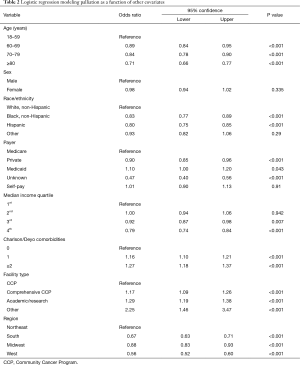
Full table
Survival analyses
Kaplan-Meier curves for the various types of palliative intervention are shown in Figure 2. After controlling for patient- and hospital-related factors using a multivariable Weibull model, palliative radiation did not significantly affect survival of patients with stage IV disease (Table 3). Both surgery [hazard ratio (HR) 0.94; 95% CI: 0.90 to 0.98] and systemic chemotherapy (HR 0.55; 95% CI: 0.53 to 0.57) were associated with improved survival. Pain management alone, combination therapy, and referrals without a designated intervention were all associated with worsened survival, when compared to patients who received neither palliative nor non-palliative interventions (Table 3). All forms of non-palliative interventions (surgery, radiation, chemotherapy, and combination therapy) were associated with improved survival (Table 3).

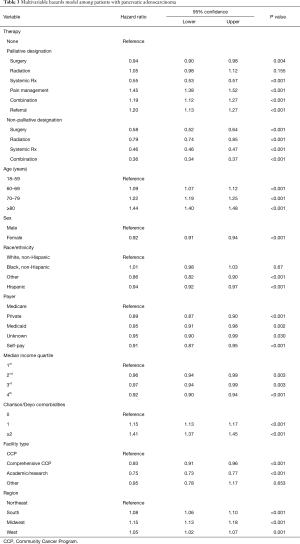
Full table
Discussion
The decision to pursue palliative therapy in patients with pancreatic adenocarcinoma is likely multifactorial, and is guided by factors such as location and character of symptoms, patient comorbidities, and patient and treating physician preferences. Focusing on a cohort of patients presenting with metastatic disease, for whom palliative therapy would likely comprise an important component of the treatment strategy, we found that that only a minority of patients (16.8%) had evidence of receiving a palliative intervention. The interventions designated as “palliative” did significantly increase during the time period, from 12.9% in 2003 to 19.2% in 2011. However, this subset still represents only a small proportion of the patients who would likely benefit from referral for palliative services. A similar trend was observed in a recent study among patients in the Netherlands, which showed an increasing proportion of patients (10% to 27%) with metastatic disease undergoing palliative chemotherapy (1). Although these observed increases are encouraging, they suggest that further efforts are necessary to promote and provide palliative therapy in this population with incurable disease.
The most common palliative modality identified in this study was systemic therapy. Pancreatic adenocarcinoma is often characterized as a systemic disease owing to its rapid infiltration and spread, limiting detection of localized disease to approximately 15–20% of incident cases (1,8). As such, treatment with systemic chemotherapy comprises the mainstay of palliative therapy for advanced pancreatic cancer (8). Multiple reports in the literature suggest a survival benefit with a variety of chemotherapy regimens, including single-agent gemcitabine or combination regiments such as FOLFIRINOX (3,8-10). Among our cohort, receipt of palliative chemotherapy was associated with prolonged survival. Aside from its modest survival benefit, a recent systematic review by Kristensen et al. highlighted its role in improving quality of life and pain scores in patients with metastatic disease (11). The goal of minimizing symptoms, either due to the disease or to the agent itself, should guide the choice of chemotherapy regimen. FOLFIRINOX has been shown to provide an incremental survival benefit over older agents, such as gemcitabine, but its higher incidence of side effects detracts from its appeal as a palliative regimen (3,10,12).
For patients with more localized symptoms, such as gastric outlet or biliary obstruction, palliation with surgical or endoscopic approaches may provide symptomatic improvement (2). Symptomatic biliary obstruction develops in approximately 80% of patients who have disease in the pancreatic head, and may cause pruritus, cholangitis, anorexia, malnutrition, or even hepatic failure (2). Surgical bypass (such as hepaticojejunostomy or choledochojejunostomy) traditionally comprised the mainstay of therapy for these symptoms. Recently, however, in accordance with the goal of minimizing therapeutic morbidity in the face of incurable disease, endoscopic therapies with stent placement are being increasingly utilized for both duodenal and biliary obstructions (2,3,13).
For gastric outlet obstruction, both palliative resection and bypass have been described to alleviate obstructive symptoms (2,3,8). Currently, palliative bypass is preferred, as a survival benefit has not been consistently shown for either approach, and bypass is associated with a lower morbidity. The risk of developing gastric outlet obstruction is so high among patients with biliary obstruction that prophylactic gastrojejunostomy is now recommended in patients undergoing palliative hepaticojejunostomy (2,3). Within our study, surgical palliation offered a small benefit and showed decreasing utilization over time, although distinction between approach (surgical versus endoscopic) and type (gastric, biliary, or both) could not be made.
Pain management forms an essential aspect of palliative care. Among patients receiving palliative interventions in our cohort, those receiving pain managements as the primary palliative therapy increased significantly from 2003 to 2011. The abdominal and back pain frequently experienced by patients with pancreatic cancer can be debilitating, and severely affect a patient’s quality of life (3). As with surgical therapy, radiation therapy can also provide palliation of localized symptoms. It is less commonly used than either systemic or surgical intervention, but can be an important adjunct to target specific areas of pain (14,15). Furthermore, although brain and bone involvement is uncommon among patients with pancreatic cancer, radiation therapy offers an important option for patients with pathologic fractures or neurologic impairment (14).
Unfortunately, our study indicates that there may be racial and ethnic disparities in providing palliative interventions to patients with unresectable pancreatic cancer. Both black race and Hispanic ethnicity were associated with lower odds of receiving designated forms of palliative care, despite controlling for demographic factors such as insurance status, income, facility type, and regional variation. This observation is not unique to our study; a recent review highlighted disparities in hospice utilization and the assessment and treatment of pain among black and Hispanic patients across a variety of advanced diseases (16). Another study utilized the Surveillance Epidemiology and End Results (SEER) database to identify cases of pancreatic adenocarcinoma and evaluate overall trends in treatment. Of the patients undergoing therapy that was determined to be palliative, patients of black race had lower odds of receiving chemotherapy, and those of Hispanic ethnicity were less likely to receive radiation (17). Although another study suggested that some of these disparities might be attenuated in the inpatient setting, it is evident that further efforts to eliminate these differences are necessary (18).
Other differences in provision of palliative therapy were noted among our study cohort. While gender does not appear to have a significant role, several other patient factors may be associated with the decision to offer palliative services. Interestingly, while patients with more comorbidities are more frequently offered palliative therapy, age had the opposite association: with increasing age, the odds of undergoing a palliative intervention decreased (Table 2). This finding was also reflected in the study by Bernards et al., which noted a lower use of palliative chemotherapy among older patients in the Netherlands (1). Socioeconomic factors also appeared to affect provision of palliative therapy among our cohort; those with higher income and private health insurance were less likely to receive palliative interventions. This finding is in distinction to that observed in the Netherlands, where those of higher socioeconomic status were more like to undergo palliative chemotherapy. Further investigation into causes of these differences is important.
This study has several limitations. We cannot be certain that the codes for palliative care are used appropriately and consistently across institutions; palliative interventions, therefore, may be underestimated in this study, although there is consistency of the estimated prevalence with other studies. Although the NCDB offers detailed staging, therapeutic, and survival data on patients with pancreatic cancer, it does not offer information regarding the specific type of chemotherapy regimens given or the type of palliative surgeries performed. Therefore, analyses comparing types of these interventions cannot be performed. Additionally, these data are subject to bias due to their retrospective nature, and the choice between various palliative interventions may be influenced by the health state of the patient. For example, the survival benefit observed with palliative chemotherapy may be in part due to the selection of healthier patients who are deemed “fit” enough to undergo chemotherapy. Furthermore, the accuracy of the “palliative” designation is dependent upon the coding by trained data entry personnel, and may be subject to substantial inter-coder variability that cannot be quantified. While any therapy in a patient with metastatic disease may be considered palliative, the lack of the palliative designation for all patients with Stage IV disease undergoing therapy indicates that the goals of therapy may not have been fully addressed with the patients during their healthcare encounters. Lastly, the database cannot discern individual patient and family preferences that may influence whether or not palliative care is pursued, and which approach is selected.
Conclusions
While the utilization of palliative therapies among patients with metastatic pancreatic cancer is increasing, deficiencies remain among a population that would likely benefit from these approaches. Moreover, there are areas of disparity—particularly among older patients and those with non-white race or ethnicity—that must be addressed on both a locoregional and national level. Since this disease is often symptomatic and can significantly impair functional status among patients who suffer from it, until medical and surgical therapies advance, the use of palliative therapies to improve quality of life among this population should remain a priority.
Acknowledgements
None.
Footnote
Conflicts of Interest: This project was presented in abstract form during a podium presentation at the European Society for Medical Oncology in Barcelona, Spain on June 29th, 2016.
Ethical Statement: This study was approved by the institutional review board of the Pennsylvania State University College of Medicine (STUDY00005949).
References
- Bernards N, Haj Mohammad N, Creemers GJ, et al. Ten weeks to live: a population-based study on treatment and survival of patients with metastatic pancreatic cancer in the south of the Netherlands. Acta Oncol 2015;54:403-10. [Crossref] [PubMed]
- Conrad C, Lillemoe KD. Surgical palliation of pancreatic cancer. Cancer J 2012;18:577-83. [Crossref] [PubMed]
- Poruk KE, Wolfgang CL. Palliative management of unresectable pancreas cancer. Surg Oncol Clin N Am 2016;25:327-37. [Crossref] [PubMed]
- DiMagno EP. Pancreatic cancer: clinical presentation, pitfalls and early clues. Ann Oncol 1999;10:140-2. [Crossref] [PubMed]
- Kuhlmann KF, de Castro SM, Wesseling JG, et al. Surgical treatment of pancreatic adenocarcinoma; actual survival and prognostic factors in 343 patients. Eur J Cancer 2004;40:549-58. [Crossref] [PubMed]
- Obermeyer Z, Makar M, Abujaber S, et al. Association between the Medicare hospice benefit and health care utilization and costs for patients with poor-prognosis cancer. JAMA 2014;312:1888-96. [Crossref] [PubMed]
- Wang JP, Wu CY, Hwang IH, et al. How different is the care of terminal pancreatic cancer patients in inpatient palliative care units and acute hospital wards? A nationwide population-based study. BMC Palliat Care 2016;15:1. [Crossref] [PubMed]
- Werner J, Combs SE, Springfeld C, et al. Advanced-stage pancreatic cancer: therapy options. Nat Rev Clin Oncol 2013;10:323-33. [Crossref] [PubMed]
- Burris HA 3rd, Moore MJ, Andersen J, et al. Improvements in survival and clinical benefit with gemcitabine as first-line therapy for patients with advanced pancreas cancer: a randomized trial. J Clin Oncol 1997;15:2403-13. [Crossref] [PubMed]
- Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med 2011;364:1817-25. [Crossref] [PubMed]
- Kristensen A, Vagnildhaug OM, Gronberg BH, et al. Does chemotherapy improve health-related quality of life in advanced pancreatic cancer? A systematic review. Crit Rev Oncol Hematol 2016;99:286-98. [Crossref] [PubMed]
- Gong J, Tuli R, Shinde A, et al. Meta-analyses of treatment standards for pancreatic cancer. Mol Clin Oncol 2016;4:315-25. [PubMed]
- Stark A, Hines OJ. Endoscopic and operative palliation strategies for pancreatic ductal adenocarcinoma. Semin Oncol 2015;42:163-76. [Crossref] [PubMed]
- Habermehl D, Brecht IC, Debus J, et al. Palliative radiation therapy in patients with metastasized pancreatic cancer - description of a rare patient group. Eur J Med Res 2014;19:24. [Crossref] [PubMed]
- Rombouts SJ, Vogel JA, van Santvoort HC, et al. Systematic review of innovative ablative therapies for the treatment of locally advanced pancreatic cancer. Br J Surg 2015;102:182-93. [Crossref] [PubMed]
- Johnson KS. Racial and ethnic disparities in palliative care. J Palliat Med 2013;16:1329-34. [Crossref] [PubMed]
- Shavers VL, Harlan LC, Jackson M, et al. Racial/ethnic patterns of care for pancreatic cancer. J Palliat Med 2009;12:623-30. [Crossref] [PubMed]
- Sharma RK, Cameron KA, Chmiel JS, et al. Racial/ethnic differences in inpatient palliative care consultation for patients with advanced cancer. J Clin Oncol 2015;33:3802-8. [Crossref] [PubMed]