Radiation induced gastroparesis—case report and literature review
Introduction
Therapeutic progress in metastatic cancers improves patient survival. During disease course, patient may benefit from several successive radiotherapy treatments. Increasingly, long-term and unusual radiation induced toxicities are described. Knowing these side effects leads to provide optimum medical care.
Gastroparesis is a syndrome caused by gastric stasis unrelated to mechanical obstruction. Main symptoms are: early satiety, nausea, vomiting, abdominal pain and bloating (1). It significantly affects quality of life (2), morbidity and mortality (3). The main etiologies are metabolic (diabetes =29%), post-surgical (13%), drugs and idiopathic (36%) (3). Other etiologies are poorly described, as Parkinson, amylose, paraneoplastic syndrome and radiotherapy.
Case presentation
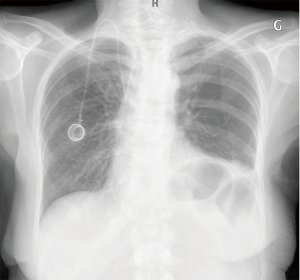
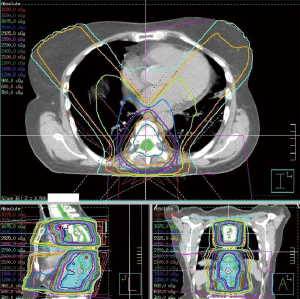
A 69-year-old woman was admitted in our department for nausea and vomiting in a context of breast cancer with bone metastases. She described nausea, abdominal fullness feeling and dysphonia (bitonal voice). These symptoms get progressively worse. After few weeks, every food consumed induced early vomiting. Chest film (Figure 1) and CT scan showed left diaphragmatic cupola rise, symptomatic of left phrenic nerve paralysis. Ear, nose and throat (ENT) examination revealed a left vocal cord paralysis due to left recurrent laryngeal nerve lesion. Gastroscopy revealed a gastric bezoar. An upper gastrointestinal (GI) series test showed an aspect of gastroparesis with gastroesophageal reflux. On examination, her medical treatment wasn’t described as causing gastroparesis. She wasn’t diabetic and hadn’t undergone recent surgery. On the other hand she underwent two sequential rachis radiotherapy (D3–D6 then D8–D11), both at the dose of 30 Gray in 10 fractions, 2 years and 7 months ago respectively (Figure 2). Cerebral MRI, plasmatic neuronal antibody and lumbar puncture were normal. Eventually we concluded to a radiation induced gastroparesis. Treatment with Domperidone was initiated without benefit after 4 weeks. Second-line treatment by intravenous erythromycin 250×3/J during 7 days and then orally over 4 weeks allowed significant reduction of GI symptoms. The patient recovered a normal nutritional status.
Discussion
Only three cases of radiation induced gastroparesis have been reported in the literature, (Table 1) (4-6). There is to our knowledge no incidence data of autonomic nervous system radiation induced neuropathy. However, many clinical presentations of late radiation-induced neuropathies have been described.
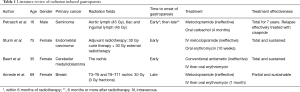
Full table
Risk factors for developing radiation induced neurotoxicity are: concomitant radiotoxic treatments, cardiovascular risk factors, length of nerve irradiated and dose per fraction.
In this case, first dose prescribed to the spine was 30 Gy in 10 fractions of 3 Gy corresponding to an equivalent 2 Gy dose (EQD2) equal to 37.5 Gy (with an alpha/beta ratio equal to 2). A second similar dose was prescribed two years later on a nearby area. Dosimetric reconstruction showed little theoretical radiation fields overlap. However, positional uncertainties as well as morphological changes of degenerative rachis may have increased the radiation fields’ superposition.
It is conventionally admitted that late radiation induced neuropathy is caused by direct nerve damage (axons, myelin and Schwann cells) or indirect lesions (ischemia, peripheral fibrosis). In this case, the dose to the stomach muscular wall was insignificant. In addition, involvement of other concomitant nerve tracts (left phrenic nerve and recurrent) suggests that a left vagus nerve lesion was the cause of digestive symptoms.
Paraneoplastic syndrome remains the principal differential diagnosis in this context. Although its exact pathogenesis remains unknown, patients’ autopsy shows neural ganglia-free stomachs, and gastric nerve fibers injured by chronic inflammation. Serological detection of anti-Hu antibodies suggests an autoimmune destruction of stomach neural plexus as a key mechanism (7). This tends to make us think that paraneoplastic gastroparesis is an intrinsic disease of the stomach. Conversely, our case was probably due to a vagus nerve lesion. These assumptions seem to be confirmed by practice. Indeed, the paraneoplastic gastroparesis cases are resistant to prokinetic drugs whereas in our case erythromycin was efficient.
Conclusions
Anatomical and chronological correlation of lesions, concomitant nerve tract damage in the radiation field and elimination of the main differential diagnosis allowed us to link this case of gastroparesis with the background of radiotherapy. This specific diagnosis led to a successful treatment and a quality of life improvement.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
Informed Consent: Written informed consent was obtained from the patient for publication of this manuscript and any accompanying images.
References
- Camilleri M, Bharucha AE, Farrugia G. Epidemiology, mechanisms, and management of diabetic gastroparesis. Clin Gastroenterol Hepatol 2011;9:5-12; quiz e7.
- Talley NJ, Young L, Bytzer P, et al. Impact of chronic gastrointestinal symptoms in diabetes mellitus on health-related quality of life. Am J Gastroenterol 2001;96:71-6. [Crossref] [PubMed]
- Jung HK, Choung RS, Locke GR 3rd, et al. The incidence, prevalence, and outcomes of patients with gastroparesis in Olmsted County, Minnesota, from 1996 to 2006. Gastroenterology 2009;136:1225-33. [Crossref] [PubMed]
- Sturm A, von der Ohe M, Rosien U, et al. Treatment of radiotherapy-induced gastroparesis with erythromycin. Dtsch Med Wochenschr 1996;121:402-5. [Crossref] [PubMed]
- Petrasch S, Layer P, Wehr M, et al. Recurrent gastroparesis following abdominal irradiation. Therapy with cisapride. Z Gastroenterol 1989;27:739-42. [PubMed]
- Baert D, Burvenich P, Lagae J, et al. Radiotherapy-induced gastroparesis: an unusual complication. J Clin Gastroenterol 2002;34:188-9. [Crossref] [PubMed]
- Donthireddy KR, Ailawadhi S, Nasser E, et al. Malignant gastroparesis: pathogenesis and management of an underrecognized disorder. J Support Oncol 2007;5:355-63. [PubMed]