Rectal cancer restaging after neoadjuvant chemoradiation: towards a down-staging score system
We have read with great interest the article of Singhal et al. (1) and we agree with their conclusions. It is our policy to submit the patients affected by rectal cancer, T3 and T4, independently from N, to a neoadjuvant treatment with 50 Grays and FOLFOX chemotherapy. Surgery follows after 4–5 weeks, and it is preceded by a restaging procedure. In order to classify the down-staging of rectal tumors, we have adopted a score system (Table 1), inspired to the current TNM classification and based on imaging criteria, usually acquired with the common contrast-enhanced radiological tools, such as computed tomography (CT) and magnetic resonance (MR). We have also considered that tumor regression follows a preferential centripetal way, starting from its most peripheral zones toward the centre of the mass and to the site of deeper penetration. We have evaluated as preeminent two main aspects of the tumor, which can condition therapeutic strategies: the involvement of rectal walls and the involvement of mesorectum. The regression of a possible secondary lympho-adenopathy was included into the concept of ‘mesorectal regression’, so avoiding the sometimes difficult distinction between secondary neoplastic nodules and metastatic lymph-nodes. Moreover, those cases with extra-fascial mesorectal diffusion were not considered in our classification, necessitating of more sophisticated examinations, such as positron emission CT, and of different neo-adjuvant treatments.
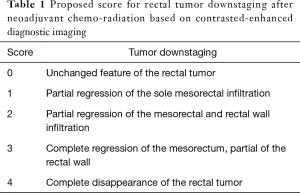
Full table
We have applied the proposed classification in 38 patients, 29 men and 9 women, aged between 60 and 75 years, observed at our institution in the period 2012–2015, where a middle or low rectal cancer had been diagnosed.
We have compared the results of our classification with the histology of rectal cancers, which was simply sub-divided into ‘high-grade’ (27 cases =71.05%) and ‘low-grade’ (11 cases =28.94%). In this way the impact of inter-observers’ diagnostic variability and intra-tumoral histological variations was abolished. Similarly, we have considered significant a down-staging corresponding to grades 3 and 4 (25 cases =65.78%), and insignificant when matching with grades 0–2 (13 cases =34.21%). In all the 11 low-grade cancers, we have found a significant down-staging; this percentage dropped to 19.28% in the 27 patients with high-grade tumors. It is evident an inverse correlation between histological severity and responsiveness to chemoradiation.
On the contrary, considering the Dworak’s classification of tumor histological regression (2), significant down-staging after chemo-radiotherapy, corresponding to grades 3 and 4, has been found in 23 cases (61%), while an insignificant down-staging, always matched with grades 0 and 1, in the remaining 15 cases. Therefore, the down-staging score appears inversely correlated with the histological grading of the tumor, but directly with the Dworak’s tumour regression.
In our classification CT and MR have a prominent role, being these tools widely used, even if we acknowledge that sometimes a neoplastic infiltration of rectal walls or mesorectum can pass unobserved at the radiological imaging, or be mistaken with a secondary fibro-inflammatory reaction to chemoradiotherapy. Correspondingly, the difference between metastatic and simply reactive lymph nodes can be sometimes difficult to ascertain.
The tumor down-staging classification helps in selecting or confirming a surgical strategy, in evaluating the risk of tumor recurrence, and in scheduling the post-operative follow-up (3-9). Our proposed classification demands to be further validated in larger cohorts of patients, and to be integrated with other aspects of rectal cancer, regarding its immunology and molecular biology.
Acknowledgements
None.
Footnote
Conflicts of Interest: The authors have no conflicts of interest to declare.
References
- Singhal N, Vallam K, Engineer R, et al. Restaging after neoadjuvant chemoradiation in rectal cancers: is histology the key in patient selection? J Gastrointest Oncol 2016;7:360-4. [Crossref] [PubMed]
- Dworak O, Keilholz L, Hoffmann A. Pathological features of rectal cancer after preoperative radiochemotherapy. Int J Colorectal Dis 1997;12:19-23. [Crossref] [PubMed]
- Ayez N, Alberda WJ, Burger JW, et al. Is restaging with chest and abdominal CT scan after neoadjuvant chemoradiotherapy for locally advanced rectal cancer necessary? Ann Surg Oncol 2013;20:155-60. [Crossref] [PubMed]
- Pomerri F, Pucciarelli S, Maretto I, et al. Prospective assessment of imaging after preoperative chemoradiotherapy for rectal cancer. Surgery 2011;149:56-64. [Crossref] [PubMed]
- MacGregor TP, Maughan TS, Sharma RA. Pathological grading of regression following neoadjuvant chemoradiation therapy: the clinical need is now. J Clin Pathol 2012;65:867-71. [Crossref] [PubMed]
- Evans J, Patel U, Brown G. Rectal cancer: primary staging and assessment after chemoradiotherapy. Semin Radiat Oncol 2011;21:169-77. [Crossref] [PubMed]
- De Nardi P, Carvello M. How reliable is current imaging in restaging rectal cancer after neoadjuvant therapy? World J Gastroenterol 2013;19:5964-72. [Crossref] [PubMed]
- Wilkins S, Haydon A, Porter I, et al. Complete Pathological Response After Neoadjuvant Long-Course Chemoradiotherapy for Rectal Cancer and Its Relationship to the Degree of T3 Mesorectal Invasion. Dis Colon Rectum 2016;59:361-8. [Crossref] [PubMed]
- Peng JY, Li ZN, Wang Y. Risk factors for local recurrence following neoadjuvant chemoradiotherapy for rectal cancers. World J Gastroenterol 2013;19:5227-37. [Crossref] [PubMed]


